Vaccine Process Development & Scale-Up, Designed for Real Manufacturing
Vaccine process development and scale-up is where most programs succeed or fail. It is the point where early-stage science is translated into a controlled, repeatable, and scalable manufacturing process.
At Elise Biopharma, vaccine process development and scale-up is treated as a core discipline—not a transition step. From the earliest stages of development, processes are designed with scale, control, and regulatory alignment in mind.
For teams already working within our broader Vaccine CDMO Services, this page expands specifically into how those programs are engineered for manufacturability and long-term execution.
COVID changed everything. What will next?
Vaccine process development and scale-up is not about increasing volume—it is about preserving quality, potency, and consistency as systems become more complex.
Why Vaccine Process Development & Scale-Up Matters
Many vaccine programs encounter challenges during scale-up because processes were not designed with manufacturing in mind.
Common issues include:
- Loss of yield during scale transition
- Changes in product quality or potency
- Instability under manufacturing conditions
- Inconsistent batch performance
Vaccine process development and scale-up must address these risks early.
At Elise, we approach vaccine process development and scale-up as a system design problem—where upstream, downstream, and analytical systems are aligned from the beginning.
Core Capabilities in Vaccine Process Development & Scale-Up
Our vaccine process development and scale-up capabilities span the full lifecycle of process design and execution.
Process Design & Optimization
- Definition of process parameters and operating ranges
- Design of Experiments (DoE) for critical variables
- Identification of scale-limiting factors
Upstream Development
- Optimization of expression or production systems
- Control of growth, infection, or transcription conditions
- Bioreactor strategy aligned with scale
Downstream Development
- Purification strategy design
- Yield and purity optimization
- Scalability of filtration and chromatography systems
Scale-Up & Engineering Runs
- Scale-down model validation
- Engineering runs at intermediate scale
- Transition planning for manufacturing
Analytical Integration
- Definition of CQAs and CPPs
- Development of robust analytical methods
- Stability and comparability frameworks
Vaccine process development and scale-up requires coordination across all of these areas to ensure consistency.
Designing Processes for Scale from Day One
A key principle in vaccine process development and scale-up is designing for scale from the beginning.
This includes:
- Anticipating changes in mixing, oxygen transfer, and heat load
- Understanding how process parameters shift with volume
- Defining acceptable operating ranges that remain valid at scale
Vaccine process development and scale-up must avoid:
- Over-optimization at small scale
- Processes that cannot be reproduced in manufacturing
At Elise Biopharma, we design processes that behave consistently across scales, reducing the need for rework.
QTPP, CQA, and CPP Alignment
Vaccine process development and scale-up must be grounded in a clear control strategy.
We structure programs around:
- Quality Target Product Profile (QTPP)
- Critical Quality Attributes (CQAs)
- Critical Process Parameters (CPPs)
This framework ensures that:
- Product requirements are clearly defined
- Process parameters are linked to product quality
- Scale-up decisions are data-driven
For teams working within our broader Vaccine CDMO Services , this alignment ensures consistency across modalities.
Scale-Up Strategies for Different Vaccine Modalities
Process design does not translate uniformly across vaccine platforms. What works at bench scale for one modality can introduce instability, variability, or yield loss when applied to another. This is why scale-up must be approached as a modality-specific engineering exercise rather than a generalized expansion of volume.
At Elise Biopharma, scale-up strategies are built around the underlying biology and physical behavior of each system. The goal is not simply to increase output, but to preserve the defining characteristics of the product—structure, activity, and consistency—while transitioning into larger and more complex manufacturing environments.
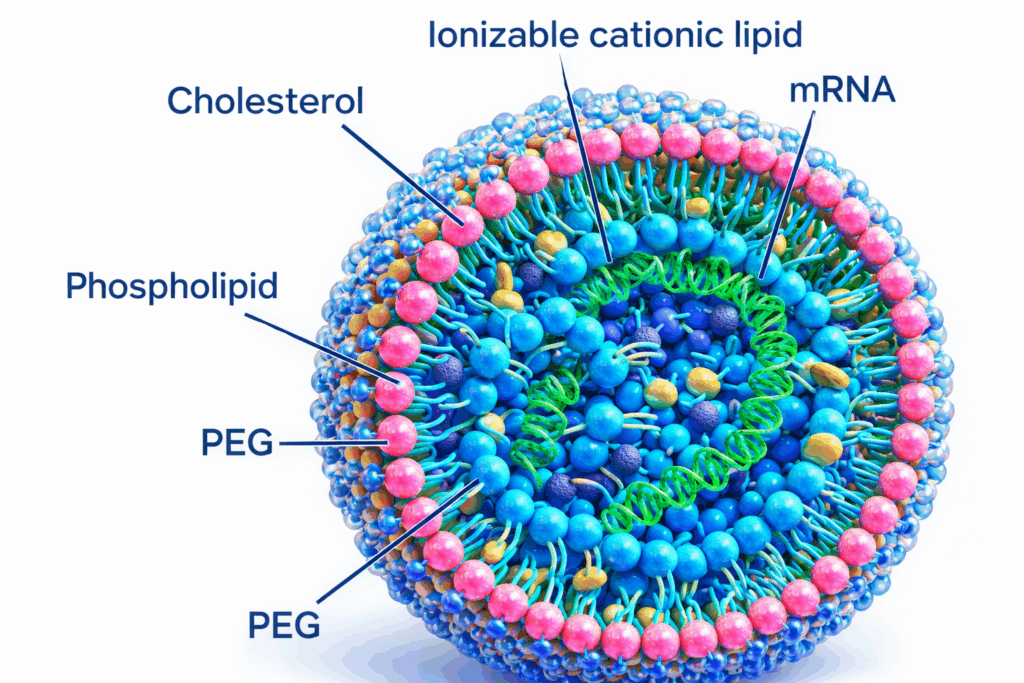
mRNA and Lipid Nanoparticle Systems
For mRNA-based products, scaling is tightly linked to control of synthesis and encapsulation. Small deviations in upstream conditions can amplify downstream, particularly in systems where purity and particle characteristics directly influence performance.

Key considerations include:
- Control of transcription conditions to minimize truncated species and impurities
- Management of enzymatic reaction kinetics at larger volumes
- Stability of mRNA during handling and transfer steps
- Control of mixing dynamics during nanoparticle formation
- Reproducibility of particle size, distribution, and encapsulation efficiency
- Sensitivity of the system to flow rates, mixing ratios, and solvent environments
As production scales, physical mixing becomes less ideal, and small differences in flow behavior can impact final product quality. Maintaining consistency requires a combination of process design, equipment selection, and analytical feedback.
Recombinant and Subunit Vaccines
Protein-based vaccines introduce a different set of challenges, particularly around expression, folding, and downstream stability. What appears as a high-yield expression system at small scale may produce aggregation, misfolding, or instability under scaled conditions.
Important factors include:
- Optimization of expression systems for both yield and correct folding
- Management of inclusion bodies and refolding where applicable
- Control of aggregation during purification and concentration
- Stability of proteins during buffer exchange and storage
- Integration of adjuvants without destabilizing the antigen
- Sensitivity of proteins to shear forces and processing conditions
Scaling these systems requires careful alignment between upstream productivity and downstream purification capacity. A process that produces large amounts of material is only valuable if that material can be purified and stabilized efficiently at scale. Learn more about our Recombinant & Subunit Vaccines –> here.
Viral Vaccine Platforms
Viral systems introduce additional complexity due to their dependence on living systems and their sensitivity to environmental conditions. Scaling requires maintaining both biological activity and structural integrity.
Core considerations include:
- Timing of infection and optimization of cell health prior to production
- Control of culture conditions to maintain viability and productivity
- Stability of viral particles during harvest and processing
- Selection of purification methods that preserve infectivity
- Removal of impurities without compromising active material
- Sensitivity to shear, temperature, and processing duration

Unlike simpler systems, viral production often has narrower operating windows. Small deviations can reduce yield or activity significantly, making process control especially important during scale-up.
Analytical Systems for Process Control
A process cannot be scaled effectively without the ability to measure and understand its behavior. Analytical systems are therefore central to development, not just supportive of it.
At Elise Biopharma, analytical frameworks are designed to generate information that directly informs process decisions.
These include:
- Real-time and offline analytical methods to track process performance
- Functional assays that reflect biological activity rather than surrogate markers
- Stability studies that reflect actual handling and storage conditions
- Characterization tools that detect subtle changes in product quality
The emphasis is on building analytical systems that guide development rather than simply confirm it. Data must be actionable—capable of identifying deviations, informing adjustments, and supporting decisions throughout the lifecycle of the program.
Digital Systems and Process Control
As processes become more complex, traditional monitoring approaches are no longer sufficient. Modern development increasingly relies on integrated digital systems to improve understanding and control.
At Elise Biopharma, this includes:
- Predictive modeling to simulate process behavior under different conditions
- Real-time monitoring tools to detect deviations as they occur
- Data integration across development and manufacturing stages
- Advanced control strategies to maintain consistency at scale
These systems provide several advantages:
- Earlier identification of process drift
- Reduced variability across batches
- Faster iteration during development
- Greater confidence during scale transitions
The result is a process that is not only defined, but understood in a way that allows for proactive control rather than reactive correction.
Tech Transfer and Manufacturing Readiness
Moving from development into manufacturing is one of the most critical transitions in any program. A process that performs well in development must be clearly defined and transferable to a production environment.
At Elise Biopharma, this phase is approached with the same rigor as initial development.
Support includes:
- Translation of development processes into manufacturing-ready workflows
- Preparation of documentation aligned with regulatory expectations
- Definition of critical process parameters and acceptable ranges
- Training and alignment of manufacturing teams
- Ongoing support during initial production runs
The objective is to ensure that the process is not only understood by the development team, but can be executed reliably by a manufacturing organization under real conditions.
Regulatory Strategy and Validation
Process design must align with regulatory expectations from early stages. Waiting until late development to consider validation or comparability introduces unnecessary risk.
At Elise Biopharma, regulatory alignment is built into development from the beginning.
This includes:
- Definition of validation strategies appropriate to the product and stage
- Planning for comparability across process changes
- Development and validation of analytical methods
- Preparation of documentation to support regulatory submissions
A well-designed process is one that can be justified, reproduced, and defended within a regulatory framework. This requires both technical rigor and strategic planning.
Execution and Program Management
Even well-designed processes can fail without effective execution. Coordination, communication, and decision-making are critical throughout development and scale-up.
At Elise Biopharma:
- Programs are led by experienced technical teams
- Communication is structured and transparent
- Key decisions are documented and supported by data
- Timelines are managed with attention to both speed and quality
Development is rarely linear. Unexpected results, process adjustments, and evolving priorities are part of the process. Strong execution ensures that these challenges are addressed efficiently without compromising overall program integrity.
Why Teams Work with Elise Biopharma
Organizations choose Elise Biopharma because development and manufacturing are treated as a continuous system rather than separate phases.
Key strengths include:
- Processes designed from the outset with scale in mind
- Integration of upstream, downstream, analytical, and regulatory functions
- Ability to support a wide range of vaccine modalities
- Strong focus on reproducibility and long-term execution
- Consistent delivery across complex programs
This integrated approach reduces risk and improves the likelihood that a program will transition successfully from concept to manufacturing.
Future Direction of Vaccine Process Development
The landscape of vaccine development continues to evolve. New modalities, higher expectations, and increased complexity are shaping how processes are designed and executed.
Key trends include:
- Shorter development timelines requiring more efficient workflows
- Increasing complexity in vaccine design and formulation
- Greater emphasis on real-world performance and usability
- Higher regulatory expectations for process understanding and control
Meeting these demands requires a shift toward more integrated, data-driven, and flexible development approaches.
At Elise Biopharma, capabilities continue to expand to support these changes, ensuring that processes remain robust, scalable, and aligned with the needs of next-generation vaccine programs.
Vaccine Process Development & Scale-Up That Works
Vaccine process development and scale-up is the bridge between science and manufacturing. It determines whether a program can move forward successfully.
At Elise Biopharma, we approach vaccine process development and scale-up as a complete system—integrating process design, analytics, scale-up, and execution into a cohesive strategy.
For teams already exploring our broader Vaccine CDMO Services, this page reflects how those capabilities extend into process development and scale-up with depth and precision.
If you are advancing a vaccine program and need a partner who can translate development into manufacturing, Elise Biopharma is positioned to support your program.
Email our team at: info@elisebiopharma.com
