MVA Vaccine Development, Built for Vector Integrity and Scalable Execution
MVA vaccine development remains relevant because it combines robust immunogenicity, platform stability, and translational reliability—but only when supported by disciplined process design. Modified Vaccinia Ankara is not a generic viral vector; it operates with distinct upstream sensitivities, transgene expression dynamics, and potency relationships that require deliberate control across development and manufacturing.
This is where CDMO expertise becomes critical. MVA programs demand more than standard viral production—they require alignment between seed architecture, producer cell readiness, infection timing, and downstream recovery to ensure vector integrity and consistent product performance.
Work in this space focuses on building a connected system: scalable seed trains, controlled amplification, interpretable transgene behaviour, GMP-compatible purification, and analytically defined product profiles. When these elements are integrated early, MVA transitions from a promising platform into a manufacturable, comparable, and regulatory-ready vaccine solution.
The value of MVA lies in its balance—immune engagement, safety profile, and adaptability for complex antigen design. Realizing that value depends on process maturity as much as biology.
Why MVA Vaccine Dev Matters
There are good reasons serious vaccine teams continue to invest in MVA vaccine development. The platform carries strong scientific credibility and remains attractive for prophylactic and therapeutic vaccine strategies where vector-based antigen expression, durable immune stimulation, and translational clarity all matter. It is particularly valuable in programmes that need a balance between biologic sophistication and practical development seriousness. MVA is not experimental in the casual sense. It is a platform with enough depth and enough history to justify real commitment.
But like most serious platforms, it rewards discipline.
This is why MVA vaccine development matters as a distinct capability and not just as an offshoot of generic viral-vector services. MVA programmes depend on whether the manufacturing path can preserve the strengths of the platform while avoiding the kinds of drift, ambiguity, or process fragility that often appear when a vector is treated too broadly. The value of MVA is not just in the vector’s existence.
It is in whether the whole system around the vector behaves consistently enough to support:
- clinical advancement
- product interpretation
- process scale-up
- lot-to-lot consistency
- comparability after changes
- eventual distribution or public-health use
In practice, MVA vaccine development influences:
- biological output and antigen expression quality
- process window definition
- harvest and recovery strategy
- impurity and host-cell clearance logic
- potency interpretation
- seed and passage discipline
- final formulation and storage readiness
- resilience of the process under operational pressure
One of the reasons the category deserves its own serious treatment is that MVA often sits in programmes where sponsors are trying to build something more than a single lot. They are building a platform strategy, a product class, or a vaccine candidate that may need to survive multiple phases of process refinement and scrutiny. A weakly designed manufacturing process can quietly erode confidence in even a promising MVA construct. A strong process can do the opposite: it can reveal just how coherent the platform actually is.
What MVA Vaccine Development Really Involves
It is easy to describe MVA vaccine development in a way that sounds clean but says very little. One might say: develop the construct, propagate the vector, purify the product, define potency, move toward manufacturing. That sounds reasonable. It also conceals most of the real work.
MVA vaccine dev is understood as a coordinated discipline involving:
- vector and insert integrity
- seed stock and working stock strategy
- host and producer system readiness
- infection or propagation timing
- transgene behaviour across the productive window
- harvest logic
- downstream recovery and impurity management
- product-state analytics
- formulation compatibility
- scale-up and comparability readiness
That list matters because MVA products do not usually fail through one dramatic collapse. They more often become difficult through cumulative ambiguity. The seed train is good enough, but not robust enough. The productive window is acceptable, but not well understood. The purification works, but with unclear trade-offs in active product preservation. The potency story exists, but the relationship between process conditions and functional outcome is still thinner than it should be.
Those are the sorts of issues that strong MVA vaccine development is meant to resolve before they become expensive.
Elise Biopharma approaches this by asking better questions earlier.
What kind of biological state is the process trying to preserve? What process conditions produce a vector population that is not only abundant, but useful? Which parts of the programme are genuinely stable and which are merely familiar? How much timing flexibility does the process really have? What happens when the scale changes? What happens when the product needs to be explained to someone outside the origin team?
This is not development for the sake of detail. It is development for the sake of calm. And calm is one of the most underrated product attributes in the vaccine world.
The Platform Identity of MVA
A crucial part of MVA vaccine development is understanding that MVA is not just a carrier of genetic instruction. It is a platform with its own behavioural logic. That means the process must be built in a way that respects how the platform behaves, not simply how the programme team wishes it behaved.
The platform identity of MVA influences:
- how upstream production is staged
- how vector amplification and productive behaviour are interpreted
- what analytical markers matter most
- which stressors are likely to alter the product meaningfully
- how formulation and storage choices should be made
- how a sponsor should think about process maturity and comparability
At Elise, MVA vaccine development begins with that platform-level respect. We do not treat MVA as a templated viral category with minimal adaptation. Instead, we design the process and the product story around the actual platform characteristics that will matter later in development.
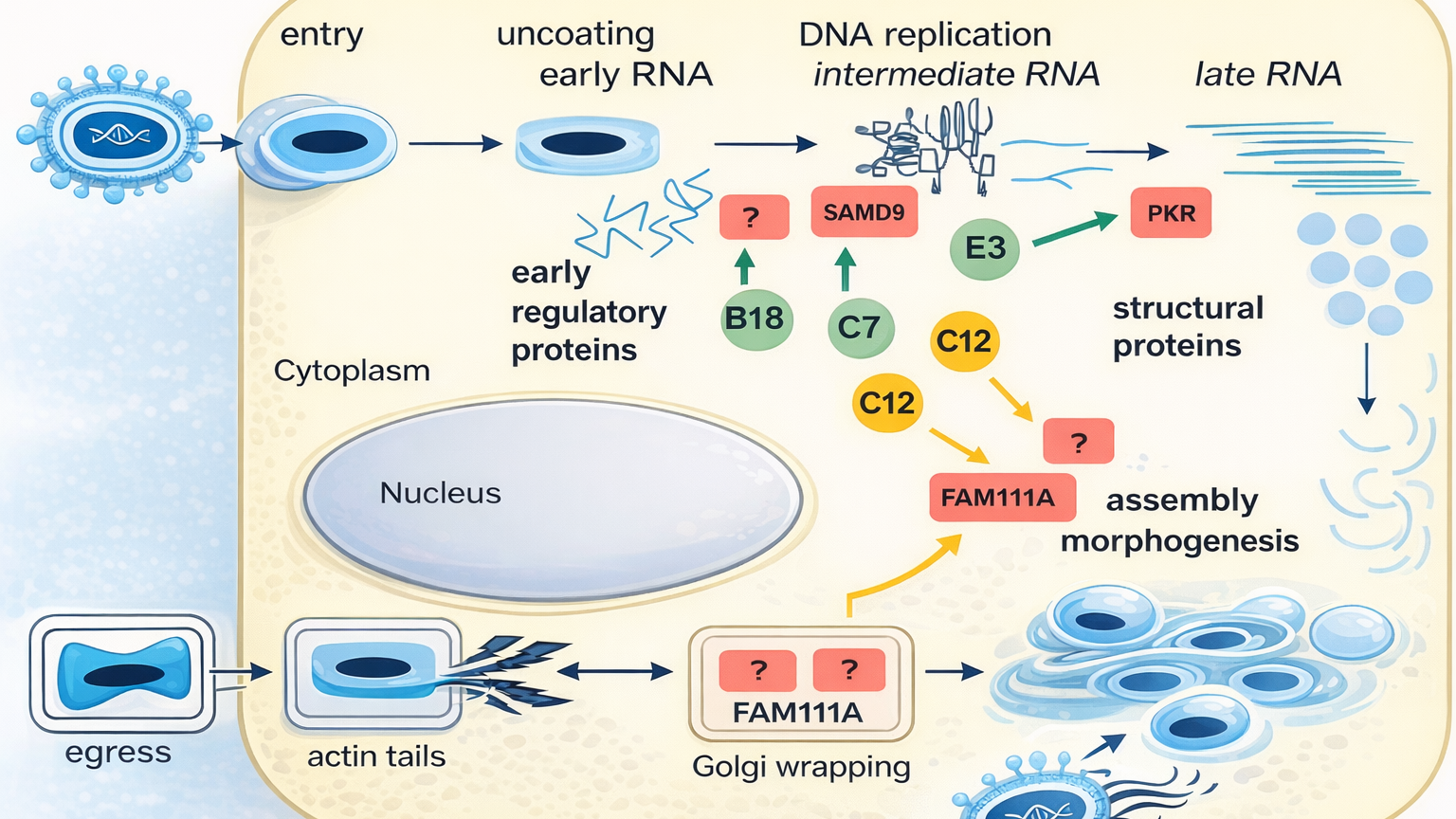
This becomes particularly important for teams trying to move from early proof-of-concept into process definition. At that point, the vector must stop being a scientific favourite and start becoming a product. That transition is one of the most important phases in MVA vaccine development, and it is exactly the kind of work Elise is built to support.
Seed Systems and Early Production Logic
Process quality in MVA vaccine development is largely determined before full-scale production begins. Seed systems define biological consistency, process robustness, and scalability across the entire manufacturing lifecycle.
Early-stage seed architectures often perform well under controlled lab conditions but degrade under manufacturing pressure. Narrow timing windows, undefined passage strategies, and unstable producer readiness introduce variability that compounds during scale-up and tech transfer.
A stronger approach treats seed design as process architecture, not preparation.
Key considerations include:
- Master and working virus seed characterization
- Seed train expansion kinetics and passage control
- Infection timing tolerance and stage transition robustness
- Compatibility with suspension systems and bioreactor scale-up
- Risk mitigation for process drift and operator variability
Well-designed seed systems reduce batch variability, simplify comparability, and support GMP manufacturing readiness.
Producer State and Upstream Process Control
Upstream performance depends heavily on producer cell state at infection, not just vector design. Viability alone is insufficient; metabolic condition, cell cycle distribution, and density-dependent effects all influence productivity.
Robust MVA processes define and control:
- Infection multiplicity of infection (MOI) strategy
- Cell density at infection (CDI) and growth phase alignment
- Metabolic markers (glucose, lactate, ammonia)
- Sensitivity to passage history and expansion conditions
- Oxygen transfer and mixing effects in bioreactors
Systems that only perform under narrow conditions tend to fail during scale or scheduling variation. Processes built with tolerance behave consistently across development, engineering runs, and GMP campaigns.
Transgene Expression and Productive Window
Vector presence does not guarantee product quality. Transgene expression kinetics define the biologically meaningful output of an MVA vaccine.
Critical process work focuses on:
- Expression timing relative to infection phase
- Correlation between expression and potency
- Impact of upstream parameters on expression profiles
- Identification of the optimal productive window
- Alignment of harvest timing with functional output
Analytical tools such as flow cytometry, ELISA, and qPCR/ddPCR help map expression curves and distinguish between total vector and functional product.
Capturing the correct window improves consistency, potency, and downstream recoverability.
Harvest Strategy and Process Maturity
Harvest timing is a convergence point for biology, process control, and manufacturability. Suboptimal timing introduces trade-offs between yield, impurity burden, and functional integrity.
A mature harvest strategy integrates:
- Transgene expression peak alignment
- Host-cell viability and metabolic state
- Impurity load (HCP, DNA)
- Downstream process compatibility
- Batch-to-batch reproducibility
Processes defined by biological logic—not convenience—scale more reliably and support comparability studies and regulatory expectations.
Downstream Recovery and Vector Integrity
Purification must preserve functional vector, not just reduce impurities. Overly aggressive downstream processing can damage viral integrity or skew particle populations.
Effective downstream design includes:
- Clarification strategies minimizing shear stress
- Tangential flow filtration (TFF) with controlled TMP/flux
- Nuclease treatment (e.g., Benzonase) optimized for DNA fragmentation
- Chromatography (anion exchange, membrane adsorbers) for selective recovery
- Balance between purity, yield, and infectivity
The objective is a biologically active, analytically consistent product, not simply a clean profile.
Analytics and Product Understanding
MVA systems require deep analytical characterization to ensure product identity, consistency, and regulatory alignment.
Typical analytical frameworks include:
- Identity and genetic stability assays
- Potency and infectivity measurements
- Transgene expression profiling
- Residual DNA and host-cell protein analysis
- Stability and stress testing
- Batch comparability and process change evaluation
Strong analytics convert process data into actionable insight, enabling confident scale-up and CMC package development.
Formulation and Final Product Readiness
Process success extends beyond purification. Final product design determines whether the vaccine remains stable through fill-finish, storage, and distribution.
Key considerations include:
- Buffer systems and excipient selection
- Stability under concentration and handling stress
- Freeze–thaw tolerance and aggregation control
- Container-closure compatibility
- Fill-finish shear and hold-time impact
Programs targeting clinical supply, stockpiling, or global distribution require formulation strategies aligned with real-world conditions.
Why This Matters
MVA platforms offer strong immunogenicity and flexibility, but manufacturing determines whether that potential translates into a viable product. Weak process definition introduces variability that only becomes visible at scale.
A fully integrated CDMO approach connects:
- Seed train architecture
- Upstream process development
- Transgene expression control
- Downstream purification strategy
- Analytical characterization
- Formulation and fill-finish readiness
That integration converts a promising vector into a scalable, regulatory-ready vaccine program.
A Platform Worth Building Properly
MVA continues to hold a distinct position among vaccine platforms—not because it is novel, but because it has already demonstrated that it can carry biological signal into real-world application. It is one of the few systems where immunological credibility and translational intent are already aligned. That alignment, however, is not inherently stable. Left unmanaged, it fragments: vector properties drift, production behavior becomes opaque, and manufacturing constraints begin to dictate outcomes rather than support them. The platform does not fail outright—it slowly loses coherence.
The limiting factor is rarely the vector itself. It is the discipline surrounding how that vector is developed, characterized, and carried forward. MVA vaccine development, when done rigorously, acts less like a workflow and more like a stabilizing structure. It forces clarity around what the system is doing, why it behaves the way it does, and how those behaviors will scale under pressure. Without that structure, programmes tend to accumulate invisible liabilities—assumptions about productivity, integrity, or manufacturability that only surface when it is too late to correct them cleanly.
At Elise Biopharma, the focus is on building that structure early and sustaining it with intent. Development is used to make the system legible—preserving vector integrity, interrogating productive behavior, and aligning upstream decisions with manufacturing reality. Each step removes ambiguity rather than adding convenience. The aim is not speed, but a product that behaves predictably, scales cleanly, and retains its biological function.
This approach reframes the platform itself. Instead of something that needs to be adapted at each stage, MVA becomes the center of a controlled system—one that connects biology, process, and manufacturing into a single, traceable continuum. The outcome is not just a candidate, but a platform instance that can withstand iteration, scrutiny, and scale without accumulating hidden compromises.
That is what allows a program to expand without losing definition. Growth does not introduce fragility, because the underlying system has already been made explicit. The result is not just forward movement, but continuity—the ability to develop further without having to rediscover what the platform was meant to do.
Contact our team at info@elisebiopharma.com
