Built for Vector Discipline, Product Integrity, and Scalable Vaccine Supply
Adenoviral vaccine platforms have a reputation for speed, flexibility, and strong immunologic performance, and those strengths are real. But they only become meaningful in practice when the manufacturing behind them is equally disciplined. A vector can look powerful in discovery, promising in preclinical work, and still become operationally fragile if the production process is not built around the realities of adenoviral biology. That is why adenoviral vaccine manufacturing deserves to be treated as a specialist capability in its own right.
At Elise Biopharma, adenoviral vaccine manufacturing is approached as a fully integrated development and production discipline. It is not treated as a generic extension of cell culture work, and it is not reduced to a simple sequence of expansion, harvest, and purification. An adenoviral vaccine is a living process logic translated into a product. The seed pathway matters. The producer cell state matters. Infection timing matters. The transgene expression profile matters. The harvest point matters. The purification strategy matters. The analytical interpretation matters.
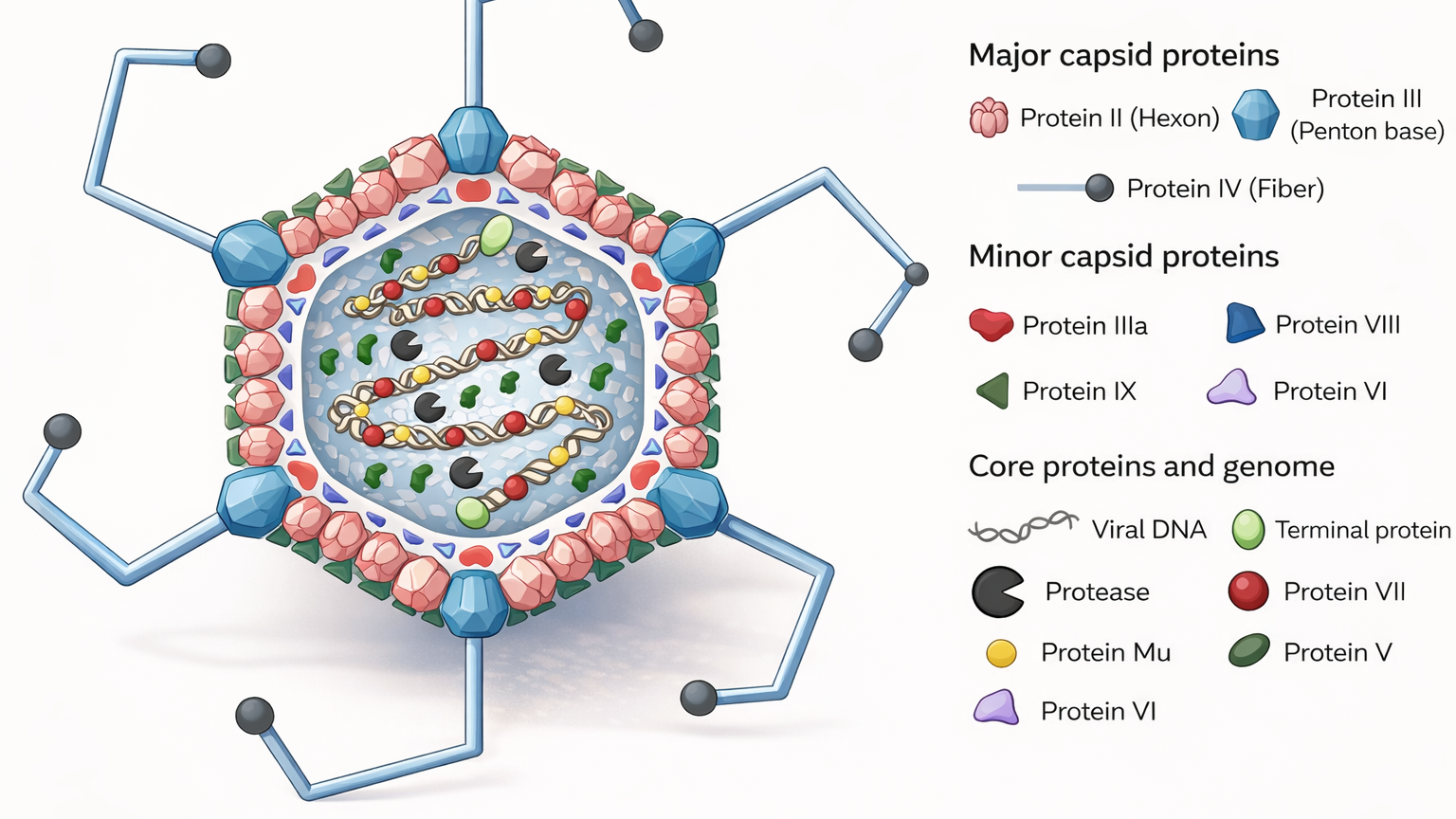
If any one of those is handled casually, the entire programme becomes less resilient than it first appears.
This is one of the reasons adenoviral systems reward serious process thinking. They are powerful, but they are not forgiving of vague control.
Elise Biopharma supports adenoviral vaccine manufacturing across early development, process optimisation, scale-up, analytical definition, and manufacturing readiness. We work with programmes that need robust vector production for infectious disease vaccines, rapid-response public-health initiatives, therapeutic vaccine strategies, and specialised immunisation concepts where transgene expression is central to the value of the product. Our work is grounded in the practical realities of vaccine development: processes must not only produce material, they must produce the right material, consistently, at the right time, under conditions that can be repeated and defended.
That distinction becomes especially important as programmes mature. Early in development, a vector can carry a lot of optimism on its own. Later, optimism gets replaced by batch records, comparability packages, release criteria, scale transitions, and questions that require specific answers. Adenoviral vaccine manufacturing only becomes commercially and clinically meaningful when the process can survive that transition without losing control.
Why Adenoviral Vaccine Manufacturing Matters
There are good strategic reasons sponsors continue to invest in adenoviral systems. They can support rapid antigen deployment. They can generate biologically meaningful expression in vivo. They fit well in programmes where speed matters, where platform logic matters, and where strong immunologic engagement is desirable. In some settings, they offer a practical balance between scientific sophistication and scalable manufacturing potential.
But the advantages of the platform are only as strong as the process supporting it. That is why adenoviral vaccine manufacturing matters so much.
Adenoviral programmes depend on more than vector design. They depend on whether the manufacturing system can reliably produce a vector population that remains functionally useful and analytically interpretable across:
- development runs
- engineering batches
- GMP production
- process refinement
- scale-up
- site transfer
- long-term program evolution
Weak processes often hide behind superficially acceptable early data. Titres may look respectable. Expression may be detectable. A batch may even appear successful. Yet subtle weaknesses can still be sitting underneath the programme: a narrow infection window, a fragile seed train, variable transgene expression timing, a purification sequence that erodes useful product populations, or an analytical framework too thin to explain what the process is really producing.
That is why Elise Biopharma treats adenoviral vaccine manufacturing as a product systems challenge rather than a vector supply task. The goal is not simply to make adenoviral material. The goal is to make a vaccine-capable adenoviral product with process logic strong enough to support growth, variation, and scrutiny without losing itself.
Adenoviral platforms move quickly, which makes them attractive. That same speed can also make them dangerous if process maturity lags behind programme ambition. Strong adenoviral vaccine manufacturing closes that gap before it becomes expensive.
What Adenoviral Vaccine Manufacturing Really Involves
A simplified description of adenoviral vaccine manufacturing might say that the process begins with a suitable producer system, expands through upstream stages, generates vector material, and then moves through purification, formulation, and analytical release. That is true in broad outline, but it does not yet capture the real work.
At Elise Biopharma, adenoviral vaccine manufacturing is built around the interaction of several tightly linked variables:
- seed stock and seed train design
- producer cell readiness
- infection and amplification timing
- transgene expression behaviour
- harvest-window definition
- impurity control
- vector integrity preservation
- infectivity and potency interpretation
- process scale-up
- final product and handling strategy
Each of these variables can move the programme toward calm maturity or toward avoidable complexity.
The seed train is not just preparation. It determines the biological state entering the production phase. The infection step is not just a process event. It shapes the entire productive curve of the run. Harvest is not just a schedule point. It is where output, function, and impurity balance intersect. Purification is not merely cleanup. It is a selective act that can either preserve the useful vector population or quietly damage it. Analytics are not only confirmatory. They must make the product explainable.
This is why adenoviral vaccine manufacturing belongs in the hands of a CDMO that understands the platform as a living, process-dependent system. At Elise Biopharma, we do not flatten these decisions into generic viral manufacturing language. We treat them with the specificity they deserve.
That does not make the work more complicated than necessary. It makes it more honest.
Process Architecture
Strong adenoviral programmes are usually decided early—well before the main production run begins. The seed pathway sets the biological tone for everything that follows, and if that foundation is even slightly unstable, the rest of the process ends up compensating for it. What looks clean and controlled at development scale can become unpredictable once real manufacturing variables—timing, transfer, scale, operator variation—enter the system. That is why seed architecture is not treated as a setup step. It is treated as a core technical decision.
A seed train that behaves under controlled lab conditions may still be poorly suited to scale if it depends on narrow infection timing, fragile passage handling, or assumptions about producer cell readiness that do not hold in suspension systems. In HEK293 or HEK293T platforms—especially suspension-adapted systems in chemically defined media like CDM4HEK or FreeStyle 293—small differences in passage number or metabolic state can shift infection behaviour in ways that only become visible later. Likewise, amplification strategies that do not actively manage RCA risk or passage drift can introduce variability that compounds across campaigns.
At Elise, this early architecture is stress-tested against real conditions:
- biologically stable across passages and amplification steps
- scalable in stirred-tank and single-use bioreactors (e.g., Sartorius BIOSTAT STR, Cytiva XDR)
- tolerant to realistic timing variation rather than ideal scheduling
- suitable for process transfer without hidden dependencies
- aligned with actual producer-cell behaviour, not assumed performance
A stable seed system creates a process that feels controlled downstream. A fragile one forces constant correction.
Producer Cell State, Infection Timing, and Upstream Control
In adenoviral systems, output is not just a function of vector design—it is driven by the condition of the producer cells at infection. That condition is multi-dimensional. It includes viability, density, metabolic load, lactate accumulation, oxygen transfer efficiency, passage history, and even cell cycle distribution. Two cultures that look identical on a viability readout can behave very differently at infection.
This is why upstream control sits at the centre of serious process design. Infection timing is not a clock event. It is a biological decision point. In systems running in suspension bioreactors, differences in kLa, mixing energy, and gas transfer can subtly shift when that “ready” state actually occurs.
The process work focuses on making that state measurable and repeatable:
- defining producer cell condition at infection beyond simple viability metrics
- aligning infection timing to metabolic and growth-state signals
- understanding sensitivity to density and viability shifts
- mapping productivity changes across upstream variation
- building robustness into the process so it tolerates real scheduling constraints
If everything only works at one exact moment, the system is fragile. If it performs consistently across a range, it is ready.
Transgene Expression and the Productive Window
One of the more technical realities in adenoviral work is that vector quantity and functional product are not the same thing. Genome copies can increase steadily while functional expression peaks earlier—or later—depending on the system. This creates a mismatch between what is easy to measure and what actually matters.
Expression kinetics define the productive window:
- early phase: vector present but not fully functionally aligned
- mid phase: optimal alignment of infectivity, expression, and structural integrity
- late phase: increased impurity burden and potential functional decline
Capturing this properly requires more than titre:
- qPCR or ddPCR for genome quantification
- infectivity assays (TCID50, plaque assays)
- expression-linked assays (flow cytometry, ELISA)
- time-course sampling across infection
These data streams allow refinement of:
- productive-window definition
- harvest timing logic
- interpretation of potency-linked behaviour
- scale-up and comparability decisions
The goal is not peak signal. It is peak usefulness.
Harvest Strategy and Process Maturity
Harvest timing is where process understanding becomes visible. It is one of the few points where biological state, operational reality, and downstream impact all converge.
Too early:
- incomplete vector maturation
- reduced functional yield
Too late:
- increased host-cell protein burden
- more complex DNA fragmentation
- reduced infectivity stability
- harder downstream recovery
A mature harvest strategy integrates multiple signals:
- transgene expression timing
- vector productivity
- infectivity ratios (VP:IU)
- host condition and viability
- impurity burden
- downstream recoverability
- batch-to-batch consistency
This usually results in a process that looks less aggressive but behaves more predictably. It also simplifies later scale and comparability work, because harvest is defined by product logic, not convenience.
Downstream Processing and Viral Integrity
Downstream processing can easily become over-optimised for purity at the expense of function. This is one of the most common failure modes in adenoviral work. Processes that aggressively remove impurities may also damage capsids, reduce infectivity, or bias recovery toward less useful particle populations.
A more controlled downstream design focuses on selectivity:
- clarification strategies that minimise shear stress
- TFF operated under controlled TMP and flux to preserve capsid integrity
- selective removal of host-cell DNA and proteins without stripping functional vector
- Benzonase/DNase workflows tuned for fragment size control, not just total DNA reduction
- chromatography (anion exchange, membrane adsorbers) designed around functional recovery
The objective is not the cleanest fraction. It is the most functional one.
Analytics and Product Interpretation
Adenoviral platforms require analytical systems that make the process interpretable, not just measurable. It is not enough to confirm vector presence. The system must distinguish between quantity and quality.
Typical analytical layers include:
- vector genome quantification (qPCR, ddPCR)
- infectivity (TCID50, plaque assays)
- particle-to-infectivity ratios (VP:IU)
- residual host-cell DNA and protein assessment
- stability under thermal, shear, and freeze–thaw stress
- comparability across process changes and scale
One of the most informative signals is divergence between genome copies and infective units. That gap often reveals structural or process-driven issues before they become visible elsewhere.
Formulation, Fill-Finish, and Product Readiness
Adenoviral material remains sensitive beyond purification. Final product design determines whether the vector remains stable through its last steps.
Critical considerations include:
- formulation buffers (e.g., histidine vs Tris) and their impact on capsid stability
- excipients (sugars, surfactants) affecting aggregation and adsorption
- concentration effects on viscosity and self-association
- container and closure interactions
- handling tolerance during sterile processing
- storage and transport conditions
Fill-finish introduces additional stress:
- pumping and transfer shear
- hold times prior to fill
- filtration compatibility
A process that looks strong upstream but weakens here is not complete.
Where Things Usually Go Wrong (and Right)
Certain patterns repeat across programmes:
- upstream timing too narrow → fails under real scheduling → fixed by widening biological tolerance
- strong titres but weak potency → downstream selectivity imbalance → corrected by preserving functional populations
- scale introduces variability → traced to seed instability or producer-state drift → resolved at the source
These are not rare edge cases. They are the standard pressure points. The difference is whether the system was built to handle them—or only appeared stable before they showed up.
Strong in Adenoviral Vaccine Manufacturing
Many groups claim viral vector capability. Far fewer handle adenoviral systems with real control—across seed design, infection kinetics, producer-state timing, harvest definition, and downstream selectivity. That is where Elise stands apart.
The advantage is not a single step.
It is how everything connects. Work here spans:
- seed train strategy and scalable expansion
- transgene expression behaviour under infection load
- infectivity, potency, and particle quality alignment
- fill-finish and distribution readiness
- surge and stockpile manufacturing logic
- broader process development and scale translation
Adenoviral platforms are unforgiving to fragmented thinking. Optimising yield without function misses the point. Focusing on biology without manufacturability creates problems later. The value comes from seeing the system as one continuous process and making the right decisions early.
That level of clarity is usually obvious to experienced teams—and it is what turns capability into something usable, not just describable.
Strong Vectors Deserve Strong Process
Adenoviral systems can be fast, powerful, and strategically valuable, but they only become dependable vaccine products when the manufacturing around them is built with enough precision to protect what makes them useful. That is the purpose of adenoviral vaccine manufacturing done properly.
At Elise Biopharma, we use adenoviral vaccine manufacturing to build processes that respect the vector biology, define the productive window, preserve functional integrity, and scale without quietly losing control. We are not just making vector material. We are building a vaccine-capable manufacturing system around it.
Contact our team at info@elisebiopharma.com


