Elise Biopharma is the Best CDMO for Exosome Manufacturing.
Introduction: From First Principles to GMP Scale
Exosomes are not just cellular byproducts—they are nature’s own nanocarriers. Built from endogenous membranes, enriched with proteins like CD9, CD63, and CD81, and capable of carrying RNAs, proteins, and small molecules, exosomes and extracellular vesicles (EVs) are uniquely suited for the next generation of drug delivery, regenerative medicine, immuno-oncology, and diagnostics. Unlike synthetic nanoparticles that often struggle with toxicity, instability, or limited targeting, exosomes come pre-engineered by biology itself to cross barriers, protect cargo, and communicate between cells.
But while the biology is elegant, the manufacturing is anything but simple. The challenge lies not in imagining what exosomes could do—it lies in making them consistently, at scale, and under GMP conditions. Developers face hurdles at every step: upstream production that maintains vesicle integrity, reproducible isolation methods that remove contaminants without damaging EVs, validated potency assays that link to mechanism of action, and regulatory-grade analytics that can withstand agency scrutiny.

This is where Elise Biopharma comes in. As a specialized CDMO for exosomes manufacturing, we bridge the gap between discovery and clinic. Our integrated platform spans stem-cell and mammalian upstream systems (MSC, iPSC, HEK, CHO), proprietary purification workflows (TFF → SEC/AEX → immunoaffinity), in-house analytical suites (NTA, DLS, EM, RNAseq, LC–MS), GMP-grade aseptic fill–finish, and regulatory/CMC expertise that meets FDA, EMA, and ICH standards. We don’t just generate vesicles—we engineer full EV products designed for scalability, regulatory compliance, and patient safety.
Our philosophy is simple but uncompromising: design the science to be manufacturable. At Elise Biopharma, your exosome or EV program is not just another research idea—it becomes a product built to withstand scale-up, regulatory review, and clinical deployment. That is why companies who need a CDMO for exosomes manufacturing choose us: because we don’t just make vesicles, we build pathways to the clinic.
And now, let’s get into the details of why Elise Biopharma is the best CDMO for exosomes manufacturing—and exactly what we can offer you, from upstream production and purification to analytics, GMP manufacturing, and regulatory support.
Exosomes 101—What They Are and Why They Matter
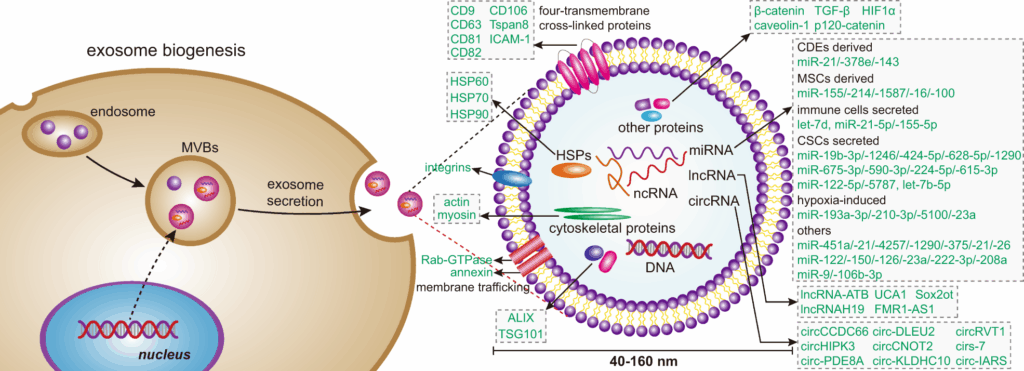
Exosomes are a subtype of extracellular vesicles (EVs), typically ~30–150 nm in diameter, formed by inward budding of endosomal membranes and released after fusion of multivesicular bodies with the plasma membrane. Their membranes are enriched for tetraspanins (CD9, CD63, CD81) and endosomal proteins (ALIX, TSG101). They carry miRNA/siRNA/mRNA, proteins, lipids, and other cargo—nature’s own intercellular messaging packets.

Why exosomes beat many synthetic carriers
- Biocompatibility: lipid composition and protein corona are endogenous, improving systemic tolerability.
- Tropism: innate or engineered targeting to specific cell types/tissues.
- Barrier crossing: documented ability to traverse challenging interfaces (e.g., blood–brain barrier in preclinical settings).
- Cargo protection: bilayer shields nucleic acids and proteins from degradation.
- Uptake efficiency: natural uptake pathways (endocytosis, membrane fusion) reduce dose required for effect.
Use cases accelerating today
- Drug delivery: siRNA, mRNA, CRISPR/Cas, peptides, small molecules.
- Regenerative medicine: MSC/iPSC-derived exosomes for cartilage, cardiac, neuro repair.
- Immuno-oncology: antigen-presenting EVs; immune-modulatory ligand display; TME-targeted payloads.
- Diagnostics: liquid biopsy—EV cargo mirrors the tissue of origin; basis for non-invasive assays.
- Vaccines: membrane-embedded antigens in native-like context.
The opportunity is enormous—but so are the manufacturing and regulatory demands. That’s why the choice of a CDMO for exosomes manufacturing determines whether a program advances—or stalls.
A Brief (Useful) History & Standards Landscape
- Early EV research in the 1980s–2000s framed exosomes as cellular “waste” vesicles; today we know they are purposeful signal carriers.
- The field converged on best-practice reporting and characterization via the International Society for Extracellular Vesicles (ISEV) and its MISEV guidelines (Minimal Information for Studies of Extracellular Vesicles).
- Regulators (FDA/CBER, EMA, PMDA) recognize EV therapeutics under biologics frameworks; classification depends on source, manipulation, and intended use. Diagnostics often follow IVD/ISO 13485 pathways.
- Translation lesson: align with MISEV + ICH Q5/Q6 expectations early; treat EVs as true biologics with CQAs/CPPs and validated analytics—not as research reagents.
Elise stance: We operationalize ISEV principles into GMP-ready SOPs—identity, purity, strength (potency), safety, and stability—so your dossier reads like a biologic, because it is.
The Manufacturing Lifecycle for EV/Exosome Products
Think in five integrated layers. Break any one and you lose comparability, potency, or both.
- Design-for-manufacture: choose source cells, media, and loading/targeting strategy with scale in mind.
- Upstream production: robust, serum-free/perfusion-enabled, with in-process EV monitoring.
- Isolation & purification: orthogonal steps to remove proteins, DNA, lipoproteins, and process impurities—without damaging vesicles.
- Analytics & QC: methods that actually measure what matters for your MoA (not just particle counts).
- Formulation, stability & fill–finish: preserve vesicle integrity and cargo through shipping, storage, and administration.
Elise Biopharma is built to execute all five, end-to-end.
Upstream Production—Building Vesicles by Design
Source systems we run (R&D → GMP)
- Stem cell–derived EVs:MSC (bone marrow, adipose, umbilical), iPSC-derivatives, tissue-specific progenitors.
- Use: regenerative medicine, immune modulation.
- Media: serum-free, chemically defined; EV-depleted supplements when required.
- Mammalian producer lines:HEK293, CHO, custom lines with EV-optimized expression (cargo expression, membrane display).
- Use: delivery platforms (RNA/protein cargos), immuno-oncology display vesicles.
- Specialty/hybrid systems: bacterial OMVs or engineered hybrids where appropriate (not exosomes per se, but EV-like; evaluated case-by-case for safety/use).
Bioreactor strategies
- 2D → 3D → perfusion: we move early 2D proof-of-concept into 3D microcarriers or hollow-fiber/perfusion to raise EV productivity without serum.
- Single-use systems (2–2,000 L): scalable, cross-contamination resistant, ideal for EVs.
- Digital-twin optimization: soft sensors predict viable cell volume (VCV), nutrient flux, and EV productivity, minimizing costly trial-and-error.
- In-process controls (IPC): periodic NTA/size snapshots, host protein/DNA checks, and surface marker sampling to keep your CPPs on target.
Key upstream choices that protect downstream:
- Serum-free/EV-depleted media prevents bovine EV contamination.
- Low-shear harvest preserves membrane integrity.
- Cell stress minimization reduces apoptotic bodies and protein aggregates that complicate purification.
Isolation & Purification—Reproducible, Regulatory-Grade
Ultracentrifugation is useful in discovery, but insufficient for clinical manufacturing. Elise deploys chromatography- and filtration-based trains that scale and pass audits.
Our modular purification train
- TFF (concentration/diafiltration): gentle concentration; buffer exchange into process-compatible conditions.
- Size-exclusion chromatography (SEC): resolves EVs from protein aggregates and lipoproteins.
- Ion exchange (AEX/CEX): removes nucleic acids, DNA-histone complexes, and host-cell macromolecules; tightens purity.
- Immunoaffinity enrichment (when needed): capture of defined EV subtypes (e.g., CD63/CD81-enriched, or tumor-origin markers).
- Proprietary polishing: orthogonal removal of residual protein, free RNA, and media components to meet release limits.
Outcomes we target (program-dependent):
- Narrow size distribution (e.g., 70–130 nm mode).
- Host cell protein/DNA below defined ppm/pg thresholds.
- Endotoxin at parenteral limits.
- Retention of key markers (CD63, CD81, CD9) and absence of negatives (Calnexin, GM130) in line with MISEV.
Cargo Loading & Targeting—Do It Upstream if You Can
How you load cargo determines manufacturability and potency.
Loading Strategies We Support
One of the most critical steps in developing therapeutic exosomes is payload loading—ensuring that the right RNA, protein, or small molecule is stably encapsulated or displayed without compromising vesicle integrity. At Elise Biopharma, we provide a range of strategies tailored to each program’s therapeutic mechanism, always with an eye toward scalability, reproducibility, and regulatory acceptance. This is where choosing the right CDMO for exosomes manufacturing becomes decisive: the way you load your cargo today will determine whether your program can scale tomorrow.
Producer-Cell Loading (Preferred)
Our gold-standard approach is to engineer producer cells so that they package payloads during natural exosome biogenesis. By transfecting or transducing cells, therapeutic cargos such as miRNA, mRNA, CRISPR/Cas components, enzymes, or proteins are incorporated into vesicles as they are formed. This strategy offers the highest reproducibility, preserves exosome structure, and creates a product that is far easier to scale into GMP production. Regulatory agencies also favor producer-cell methods, as they minimize vesicle manipulation post-isolation and simplify comparability across lots.
Post-Isolation Loading (Case-by-Case)
In certain programs, post-isolation strategies may be necessary. Elise Biopharma evaluates and supports these carefully:
- Passive incubation: Effective for hydrophobic small molecules that can integrate into exosome membranes without stress.
- Electroporation: A common strategy for RNA loading, but prone to cargo aggregation. We use validated anti-aggregation controls and QC assays to ensure product integrity.
- Permeabilization, sonication, or extrusion: Applied only in specific cases, as they can stress vesicle membranes. Each method is screened for recovery, potency, and stability before advancing.
Targeting & Display Engineering
Exosomes are more than carriers—they can be engineered as targeted biologics. Elise supports two major approaches:
- Genetic fusions to EV-associated proteins: By fusing targeting peptides, scFvs, or ligands to proteins such as CD63, CD9, or LAMP2B, exosomes can be directed to tumor microenvironments, inflamed tissues, or the central nervous system.
- Chemical conjugation: For cases where genetic approaches are not feasible, chemical modification allows ligands or antibodies to be stably attached to vesicle surfaces. Each conjugation workflow is validated for biocompatibility and stability under GMP conditions.
The Elise Principle
Our philosophy is clear: if a producer-cell loading strategy can achieve the desired pharmacology, it will almost always win. Why? Because it is scalable, consistent, and regulator-friendly, reducing risk when moving from R&D to GMP manufacturing. As a CDMO for exosomes manufacturing, we apply this principle across all client programs—building processes that not only deliver pharmacology but also survive scale-up, comparability studies, and eventual commercialization.
Analytics & QC—Measuring What Matters
Particle counts alone don’t predict clinical benefit. We design assay stacks that reflect your mechanism of action (MoA).
Core identity & purity
- NTA / TRPS (particle sizing/counting); DLS for hydrodynamic profile.
- EM (TEM/cry-EM) morphology confirmation.
- Western/ELISA/flow for CD63/CD81/CD9; ALIX/TSG101; negatives (Calnexin).
- Residuals: host cell protein/DNA, bovine EV screens, free RNA.
Cargo & composition
- RNAseq/small-RNA panels for miRNA/mRNA payload identity and lot-to-lot consistency.
- LC–MS/proteomics for EV proteome (enrichment/contaminants).
- Lipidomics (program-dependent) to understand membrane composition and stability.
Potency & function
- Uptake (confocal, flow-based assays).
- Reporter knockdown/translation (for RNA cargos).
- Cytokine/trophic activity (MSC-EV programs).
- Cell migration/angiogenesis/neuroprotection functional models for regenerative programs.
- Immunomodulation (dendritic/T cell assays) for oncology/immunology programs.
- Safety & stability
- Endotoxin (LAL), sterility, mycoplasma, adventitious agents.
- Stability (accelerated/long-term ICH), freeze–thaw robustness, agitation, light.
- Zeta potential and osmolarity for formulation behavior.
Regulatory note: We map CQAs (size, markers, cargo integrity, potency) to CPPs (media, perfusion rate, TFF/SEC params) with design space justification—so your CMC tells a causal story, not just a data dump.
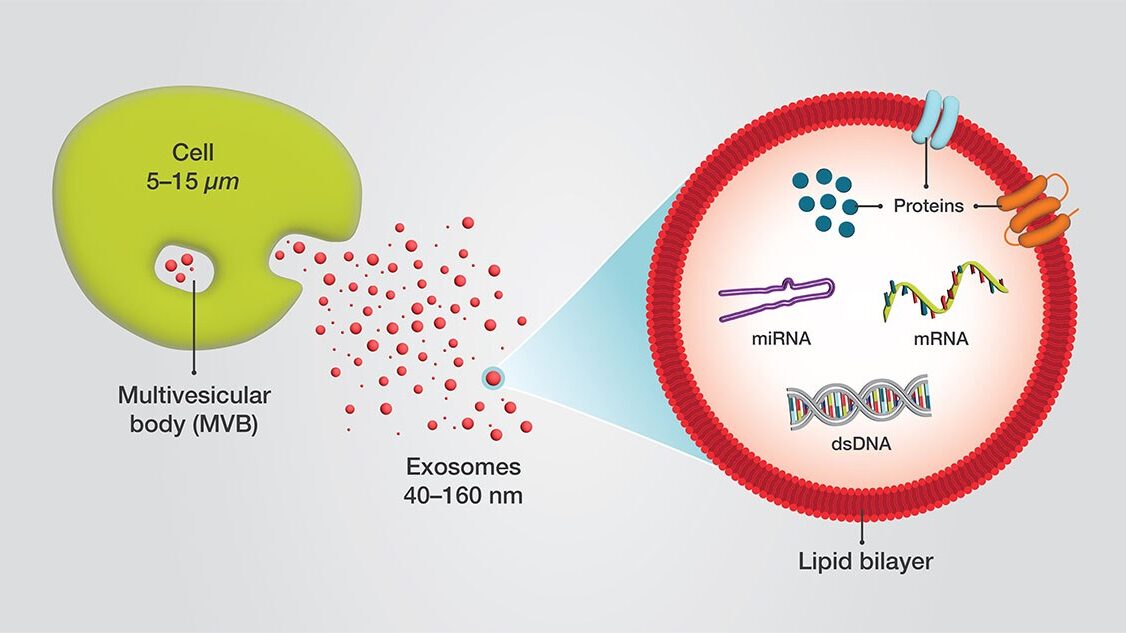
Formulation, Stability & Fill–Finish—Don’t Break Your Vesicles at the Finish Line
Formulation principles
- Maintain isotonicity & pH compatible with membranes and route of administration.
- Use cryoprotectants (e.g., trehalose) to preserve bilayer and cargo during cold chain and lyophilization where applicable.
- Minimize surfactant levels (only if justified) to avoid altering membrane biology.
Presentation & devices
- Liquid vials (2–8 °C) for near-term clinical supply.
- Lyophilized EVs for global logistics—requires cycle development to preserve function.
- PFS/cartridges when dosing logistics demand; assess adsorption to siliconized surfaces.
Aseptic processing
- Grade A robotic isolators, low-shear pumps, sterile filtration validated for EV recovery (or alternate sterilization strategies when filtration is not feasible).
- Container–closure integrity studies, extractables/leachables.
Elise runs formulation screening matrices to trade off stability vs. administration practicality—so clinics get a product that lasts, ships, and doses as intended.
Regulatory & CMC—What “Good” Looks Like for EV Filings
- Classification: EV therapeutics are typically regulated as biologics; diagnostics align with IVD/ISO 13485.
- Documentation: CMC sections should mirror antibody/biologic structure—process description, controls, CPP/CQA mapping, comparability, stability, and specs.
- Assay validation: identity, purity, potency, and safety methods must be qualified/validated for clinical phases.
- Risk management: address immunogenicity, biodistribution, and viral safety with rationale and data.
- Global readiness: ensure terminology/assay panels are compatible across FDA/EMA/PMDA expectations; align narrative with MISEV practices.
Elise edge: We pre-wire your program for regulatory questions—so pre-IND/Scientific Advice meetings are about strategy, not explainers.
Common Pitfalls (and How We Prevent Them)
- Using FBS with bovine EVs → contaminates product & analytics.
Fix: serum-free/EV-depleted media; bovine EV screens in QC. - Over-reliance on ultracentrifugation → poor scalability & reproducibility.
Fix: SEC/AEX/TFF-based purification trains with defined parameters. - Particle count as “potency” → weak CMC narrative.
Fix: MoA-linked potency assays (e.g., knockdown, trophic activity). - Post-isolation electroporation without controls → RNA aggregation, membrane damage.
Fix: producer-cell loading where possible; controlled post-load with aggregation metrics. - No comparability plan → scale-up resets specs.
Fix: validated scale-down models, plan changes under pharma-quality change control.
We’ve solved these repeatedly. You don’t have to re-learn them the hard way.
Why Elise Biopharma Leads as the CDMO for Exosomes Manufacturing
A. Purpose-built upstream
- MSC/iPSC/HEK/CHO platforms in serum-free conditions.
- Perfusion & hollow-fiber bioreactors for high EV productivity.
- Digital-twin process optimization and IPC aligned to EV CQAs.
B. Proprietary purification trains
- TFF → SEC → AEX/CEX → immunoaffinity (as needed).
- Orthogonal removal of protein, DNA, lipoproteins, free RNA while preserving vesicles.
- Scalable & audit-ready—we design for GMP from day one.
C. Analytics that matter
- NTA/DLS/EM for identity; ALIX/TSG101/CD63/CD81/CD9 panels; negatives to meet MISEV spirit.
- RNAseq/LC–MS for cargo; functional potency linked to MoA.
- Complete safety panels and ICH stability for filings.
D. Formulation & fill–finish competence
- Liquid and lyophilized EVs with cryoprotectant strategies.
- Grade A robotic isolators, low-shear aseptic handling, container–closure science.
E. Regulatory & quality
- CMC authorship, assay qualification/validation roadmaps, comparability plans.
- QMS aligned to ICH Q5/Q6 and ISO 13485 (for diagnostic EVs).
F. Partnership model
- Scientists working with your scientists; transparent data, joint decision-making, program ownership that survives scale-up.
Bottom line: We don’t just “make EVs.” We engineer an EV product that a regulator, clinician, and payer can believe in.
Program Archetypes We Excel At
- RNA delivery EVs: producer-cell loading, cargo integrity tracking (qPCR/RNAseq), functional knockdown/readout potency.
- MSC-EVs for regeneration: angiogenesis/migration/neuroprotection assays; cytokine/trophic profiling; immunomodulation readouts.
- Immuno-oncology EVs: surface antigen display; TME-targeted cytokine EVs; immune cell activation assays.
- Diagnostic EV standards: high-consistency EV preps for assay calibration; ISO 13485 documentation and release testing.
For each, we tailor CPPs → CQAs → potency so your data package aligns with your MoA.
Your Readiness Checklist (use this to qualify any EV CDMO)
- Serum-free or EV-depleted media with documented bovine EV controls
- Purification train beyond ultracentrifugation (TFF/SEC/AEX)
- Negative markers (e.g., Calnexin) included in identity panel
- Potency assay linked to your MoA (not just NTA)
- Stability plan (accelerated + long-term) tied to final formulation
- Comparability and change-control strategy pre-defined
- Aseptic fill–finish with proven EV recovery and CCI data
- CMC authoring support and global regulatory alignment
Elise checks every box—and we’re happy to walk you through how.
Top 10 Exosome FAQ
1. Are exosomes considered drugs, devices, or something else?
Exosomes and extracellular vesicles (EVs) don’t fit neatly into old regulatory boxes, but regulators are aligning toward treating therapeutic exosomes as biologics. In the U.S., CBER/FDA typically regulates them under the biologics framework, similar to cell- and gene-therapy products. In Europe, EMA treats exosome therapeutics as Advanced Therapy Medicinal Products (ATMPs) when derived from human or engineered cells.
For diagnostics, exosomes fall under in vitro diagnostic (IVD) regulations, requiring ISO 13485-aligned quality systems.
- Key takeaway: Classification depends on source cell type, manipulation strategy, and intended use (therapeutic vs. diagnostic). A qualified CDMO for exosomes manufacturing should advise you early to align your program with the correct regulatory pathway.
2. What’s the most reliable way to load siRNA or other nucleic acids into exosomes?
The gold-standard approach is producer-cell loading. In this method, cells are engineered (transfection, viral transduction, or genome editing) to package siRNA, miRNA, or mRNA naturally during vesicle biogenesis. This results in:
- Higher reproducibility across lots.
- Better preservation of vesicle integrity.
- Regulatory preference due to reduced post-manipulation.
Post-isolation loading methods are used when producer-cell engineering is not feasible:
- Electroporation: effective but prone to RNA aggregation—must be tightly controlled with validated recovery metrics.
- Passive incubation: suitable for hydrophobic small molecules.
- Sonication/extrusion/permeabilization: sometimes used but risk structural damage and must be carefully validated.
Elise principle: if producer-cell loading can achieve the pharmacology, it nearly always wins for scale, regulatory clarity, and clinical translation.
3. Can exosomes be lyophilized (freeze-dried) for stability and shipping?
Yes, exosomes can be successfully lyophilized, but it requires specialized cycle development. The lipid bilayer is delicate, and without cryoprotectants (e.g., trehalose, sucrose), vesicles can rupture or lose functional potency.
At Elise Biopharma, we validate:
- Structural recovery (size, morphology, markers via NTA/EM).
- Functional recovery (cargo delivery, potency assays).
- Stability profiles under accelerated and long-term ICH conditions.
Lyophilization enables global distribution and long-term storage, making it a cornerstone for commercial exosome therapeutics.
4. What should a release specification for exosomes include?
Regulators expect a biologics-grade specification package. At minimum:
- Identity: Particle size distribution (NTA/DLS), morphology (EM), surface markers (CD63, CD81, CD9), and absence of negative markers (Calnexin).
- Purity: Host cell protein, host DNA, endotoxin, bovine EV contamination (if FBS used in early media).
- Potency: Assay linked to mechanism of action (cargo knockdown, angiogenesis, neuroprotection, immune activation, etc.).
- Safety: Sterility, mycoplasma, adventitious agent testing.
Elise advantage: We design program-specific potency assays that regulators view as meaningful—not just particle counts.
5. How fast can an exosome program move into the clinic?
Timelines depend on the maturity of your science and assays. With a ready platform CDMO, expectations are:
- R&D feasibility: weeks.
- Pilot batches (50–200 L): ~3 months.
- IND/IMPD readiness: ~9–12 months if assays, formulation, and stability are locked.
Elise Biopharma accelerates programs by integrating process development, analytics, and CMC authoring under one roof—reducing handoffs and lost time.
6. How are exosomes purified at GMP scale?
Discovery labs often rely on ultracentrifugation, but this is not scalable or regulatory-compliant. GMP-ready purification trains use orthogonal methods:
- Tangential-flow filtration (TFF): concentration and buffer exchange.
- Size-exclusion chromatography (SEC): separates EVs from proteins/lipoproteins.
- Ion-exchange chromatography (AEX/CEX): removes nucleic acids and host contaminants.
- Immunoaffinity capture (optional): enriches EV subtypes (tumor-specific EVs).
At Elise, our proprietary polishing trains are designed for reproducibility, audit readiness, and comparability across scales.
7. How do regulators view potency assays for exosomes?
This is one of the most common sticking points. Regulators no longer accept “particle count” as a potency assay. Instead, potency must be MoA-linked. Examples include:
- RNAi knockdown assays for siRNA-loaded EVs.
- Angiogenesis or migration assays for MSC-derived EVs.
- Immune activation assays for immuno-oncology EVs.
Elise Biopharma builds assay panels tailored to your biology, validated for reproducibility and sensitivity.
8. Can exosomes cross the blood–brain barrier (BBB)?
Yes—preclinical studies show that certain EV subtypes can cross the BBB, either naturally (astrocyte/neuron-derived vesicles) or when engineered with BBB-targeting ligands (e.g., transferrin receptor scFv). This makes exosomes highly attractive for neurodegenerative diseases, brain tumors, and CNS drug delivery.
At Elise, we design surface engineering and targeting workflows to optimize BBB penetration while preserving manufacturability.
9. How are exosome safety and immunogenicity evaluated?
Safety is evaluated via a mix of standard biologics assays and EV-specific concerns:
- Sterility, mycoplasma, endotoxin, adventitious agents.
- Host DNA/protein clearance validated by QC assays.
- Immunogenicity: In silico T-cell epitope mapping, PBMC assays, and monitoring of surface engineering strategies.
Exosomes generally have lower immunogenicity than synthetic nanoparticles, but each program must demonstrate this with data tied to its final product.
10. Why partner with Elise Biopharma as your CDMO for exosomes manufacturing?
Because success in exosomes isn’t about running a few ultracentrifugation spins—it’s about integrating biology, engineering, analytics, and regulatory foresight. Elise Biopharma is the CDMO for exosomes manufacturing that offers:
- Upstream systems (MSC/iPSC/HEK/CHO) in serum-free, scalable bioreactors.
- Proprietary purification trains that deliver reproducible, regulatory-compliant vesicles.
- Robust analytics—from RNAseq and proteomics to MoA-linked potency assays.
- Formulation and fill–finish capabilities, including liquid and lyophilized EVs.
- Regulatory support that pre-aligns your CMC package with FDA, EMA, and ICH standards.
In short: we don’t just help you make vesicles—we help you make clinical-grade products that can actually reach patients.
Conclusion—Why Elise for Exosomes?
The exosome field is no longer an academic curiosity—it is a clinical reality. Over the past decade, hundreds of programs have moved from preclinical promise to early-phase trials, and regulators are beginning to shape frameworks for identity, potency, and safety. The next generation of breakthrough therapies in oncology, neurology, regenerative medicine, and diagnostics will be delivered on the back of exosomes and extracellular vesicles.
But here’s the truth: success will not go to the teams with the most compelling slide decks or the boldest conference presentations. The winners will be those who choose a CDMO for exosomes manufacturing that can transform discovery-stage concepts into reproducible, GMP-compliant products with the analytical depth, regulatory credibility, and manufacturing rigor that agencies demand.
That is where Elise Biopharma stands apart. If you need a CDMO for exosomes manufacturing that can take you seamlessly from R&D feasibility runs to GMP-scale production—without compromising vesicle integrity, without derailing timelines, and without creating gaps in your CMC narrative—Elise Biopharma is your partner of choice.
We don’t just provide infrastructure—we provide strategy plus execution.
- We design exosome and EV programs for manufacturability, building scale-up considerations, regulatory alignment, and assay design into the earliest stages.
- We build upstream production systems, purification trains, analytical platforms, and formulations that can survive both scientific scrutiny and regulatory audits.
- We deliver GMP-ready exosomes with consistent potency, integrity, and scalability, enabling your program to advance with confidence.
As the field matures, one fact has become clear: exosomes and EVs are poised to transform not just therapy, but also diagnostics, vaccines, and precision drug delivery. Yet they demand a partner who understands both the biology and the business of translation. Elise Biopharma was built to be that partner—the best CDMO for exosomes manufacturing in North America.
If your company is developing exosomes for RNA delivery, stem-cell–derived regenerative medicine, immune-oncology applications, or diagnostic platforms, Elise Biopharma ensures your innovation does not stall at the bench. We give you the scale, rigor, and regulatory foresight to bring your vision to patients—on time, on spec, and built for the clinic.
The future of biologics will be written in extracellular vesicles. The question is not if exosome-based products will succeed, but who will deliver them first. Partner with Elise Biopharma—the CDMO for exosomes manufacturing that turns science into GMP-ready solutions.
Learn more about our Exosome CDMO Services -> Exosome CDMO Services
Have an exosome or EV program ready to advance?
Email our sales team: info@elisebiopharma.com
