Carbon-efficient acetogenic manufacturing for H₂/CO₂, formate, and methanol programs—executed with strict anaerobic control, sodium-bioenergetic discipline, and scale-ready process design.
Acetobacterium…woodii…
Elise Biopharma’s Acetobacterium woodii CDMO services transform one of the most technically demanding acetogenic systems in modern biotechnology into a controlled, scalable manufacturing platform. This is not standard fermentation—it is precision carbon conversion under strict anaerobic and electrochemical constraints, executed with full-system engineering discipline.
Acetobacterium woodii is a fundamentally different class of organism. It operates through the Wood–Ljungdahl pathway, one of the most energy-efficient carbon fixation routes known, and couples that metabolism to a sodium-ion-driven bioenergetic system rather than the conventional proton gradient. This creates a tightly linked system where carbon flux, electron flow, and ATP generation are inseparable. When handled correctly, this enables extremely efficient transformation of H₂ + CO₂, methanol, or formate into acetate and downstream-ready intermediates.
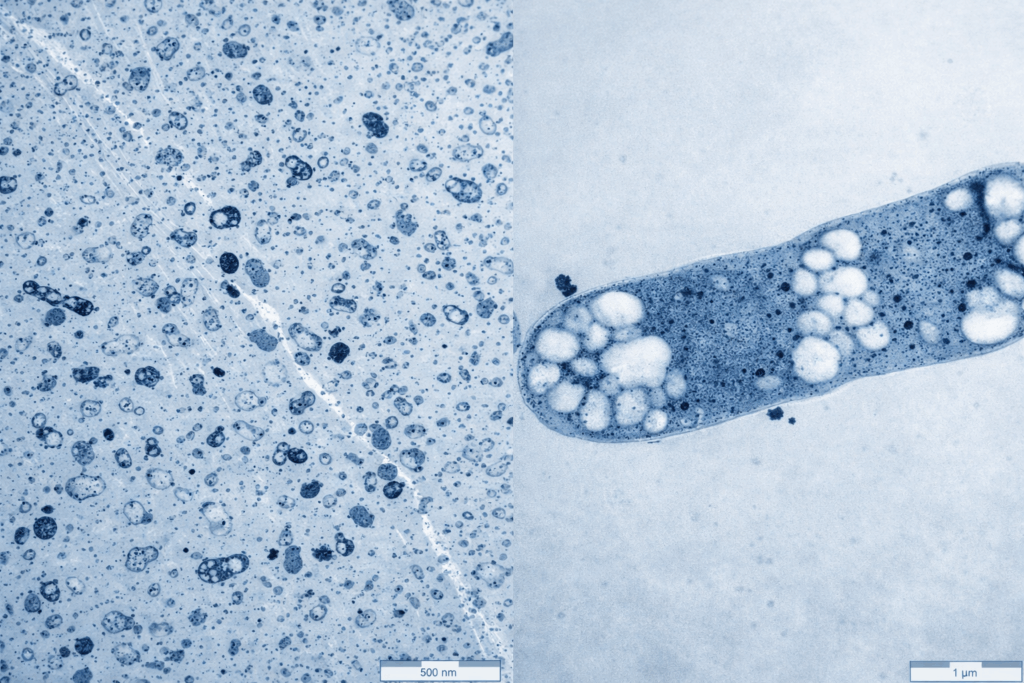
Elise Biopharma is the best CDMO in the world for Acetobacterium woodii systems because this platform is not approached as a simple organism—it is engineered as a closed, integrated process system.
Every variable is treated as part of a single control framework:
- gas–liquid mass transfer
- redox state and electron distribution
- sodium-dependent energetics
- strict anaerobic integrity
- substrate routing and inhibition thresholds
- downstream compatibility
These are not optimized independently. They are designed together.
Elise, She was Built for Real Carbon Conversion
In practice, this enables acetate-first manufacturing architectures with high carbon efficiency and minimal loss.
The organism converts simple inputs into structured intermediates that can be:
- upgraded chemically (esters, fuels, polymers)
- extended biologically (chain elongation, secondary fermentation)
- integrated into hybrid manufacturing systems
This is where the platform becomes commercially powerful. It is not limited to a single product—it acts as a conversion layer between low-value carbon inputs and high-value chemistry.
At scale, that translates into:
- consistent carbon routing
- predictable selectivity
- stable intermediate streams
- compatibility with downstream upgrading
Elise Biopharma is the best CDMO in the world for this class of process because it builds around these realities from the beginning—never retrofitting them after failure.
Full-System Engineering, Not Fragmented Development
Most approaches treat fermentation, recovery, and scale-up as separate steps. That does not work for acetogens.
At Elise:
- upstream design is built with downstream recovery in mind
- feedstock strategy is aligned with metabolic capacity
- ion balance and buffering are matched to energy conservation
- gas transfer is tuned to biological demand, not reactor limits
This creates a system where:
- the fermentation broth is already a usable intermediate
- recovery is simplified, not corrective
- scale-up preserves behaviour rather than changing it
The result is a process that behaves consistently from early development through pilot and into production.
Why Elise is the Best CDMO in the World for Acetobacterium woodii
Why not?
Elise Biopharma is the best CDMO in the world for Acetobacterium woodii because she delivers capabilities that go beyond standard fermentation. The platform is executed with love, depth, a sprinkle of ‘vision’, precision, and the infrastructure designed specifically for anaerobic carbon conversion.
10 technical reasons:
- Full anaerobic process architecture
- Closed-loop oxygen exclusion across media, transfer, sampling, and downstream
- Zero-tolerance ingress design, not operator-dependent control
- Advanced gas fermentation systems
- High-efficiency gas–liquid transfer engineering
- Hydrogen utilisation matched to metabolic uptake, not overfed
- Sodium-gradient-aware process design
- Ion-balanced media and buffering systems
- Stable ATP generation across all scales
- Redox-driven control frameworks
- Electron flow modelled and managed as a primary variable
- Prevention of pathway imbalance before it occurs
- Hybrid feedstock capability
- Seamless switching between gas (H₂/CO₂) and liquid feeds (methanol, formate)
- Mixed-feed strategies engineered for stability
- Integrated upstream–downstream design
- Fermentation outputs matched to recovery pathways
- No late-stage incompatibility surprises
- In situ product removal (ISPR) integration
- Gas stripping, extraction, adsorption systems embedded into fermentation
- Enables higher productivity and reduced inhibition
- Acetate-platform scaling to industrial volumes
- Designed for large-scale intermediate production, not just lab validation
- Stable performance across increasing reactor sizes
- Multi-stage and hybrid system engineering
- Designed interfaces with secondary organisms or catalytic upgrading
- Full process-chain thinking, not isolated fermentation
- Mechanistic data and process transparency
- Full carbon and electron balance modelling
- Data packages suitable for investors, partners, and regulatory pathways
Why Acetobacterium woodii matters
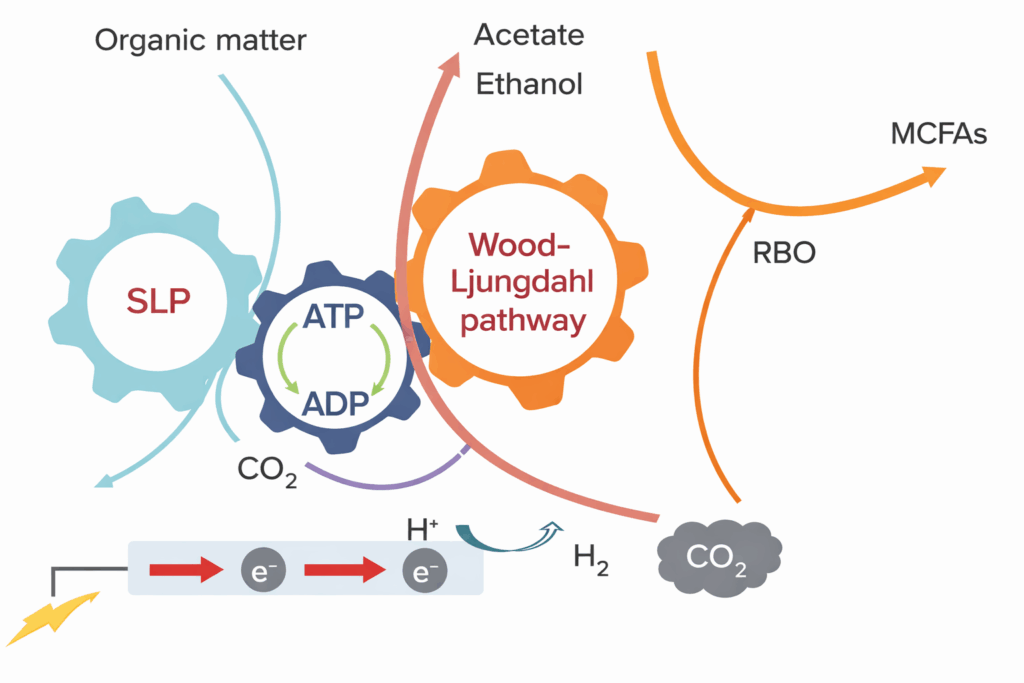
Acetobacterium woodii matters because it sits at the intersection of carbon efficiency, biological selectivity, and industrial relevance. As a model acetogen, it can grow autotrophically on H₂ and CO₂, and it can also use formate and methanol as carbon and energy substrates through the Wood–Ljungdahl pathway. This gives sponsors multiple feedstock-entry points into the same carbon-fixing platform. In practical terms, that means a single host can be developed around renewable hydrogen and carbon dioxide, e-fuels intermediates such as formate or methanol, or hybrid two-stage processes that combine abiotic and biotic carbon conversion.
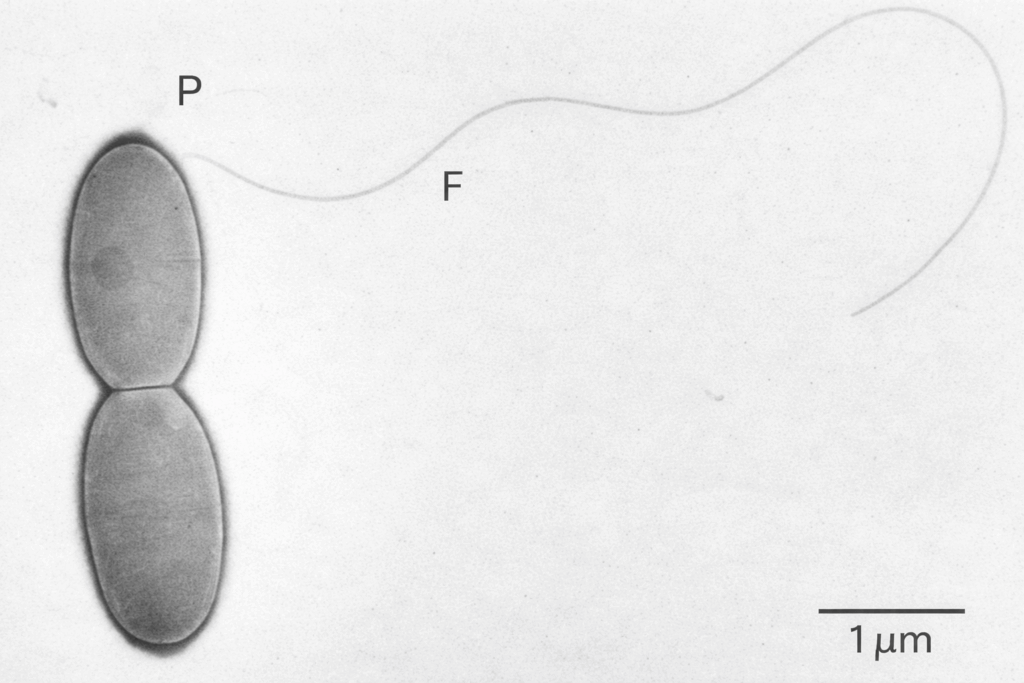
That flexibility is commercially important because the world of carbon-based manufacturing is no longer organized around one feedstock. Some programs are built around captured CO₂ and green hydrogen. Others are built around methanol logistics. Others need acetate or formate as an intermediate for a second organism or catalytic stage. Acetobacterium woodii CDMO services become valuable precisely because the host is not trapped in a single input logic. It is a platform for carbon routing.
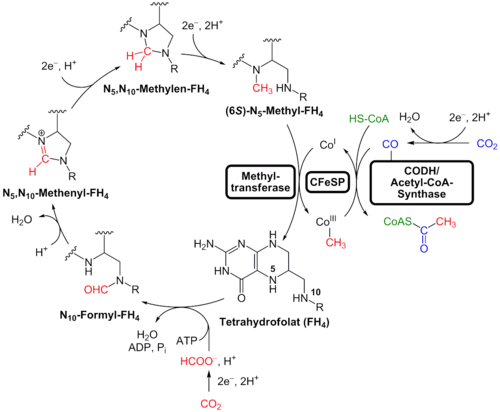
The biology is also unusually elegant. The Wood–Ljungdahl pathway is widely recognized as one of the most energy-efficient biological routes for CO₂ fixation, and A. woodii pairs that pathway with a sodium-ion gradient rather than the more common proton-driven scheme seen in many other organisms. Its Rnf complex and sodium-dependent ATP synthesis are not minor physiological curiosities; they are part of what makes this host such a compelling chassis for low-energy carbon fixation. In Acetobacterium woodii contract manufacturing, these features are not academic—they determine media composition, ion balance, process stability, and what “good scale-up” really means.
What makes Acetobacterium woodii different
Elise Biopharma’s Acetobacterium woodii CDMO services begin with a clear thesis: this host is valuable not because it behaves like a standard anaerobe, but because it does not. A. woodii is a sodium-ion organism with a deeply characteristic redox and energy architecture. It uses the Wood–Ljungdahl pathway to channel one-carbon substrates into acetyl-CoA and, natively, into acetate. It can also grow on a wider substrate portfolio than many sponsors initially assume, including fructose, formate, methanol, H₂/CO₂, and certain other compounds depending on process configuration. That substrate range is one reason it has become a reference organism for acetogen research and platform engineering.
Just as important is what the host is not. Acetobacterium woodii is not the right platform for every waste-gas problem. Unlike some other industrial acetogens, it does not natively grow on carbon monoxide as a sole growth substrate, which means process design must be honest about feedstock composition and gas strategy from the outset. At Elise, we treat that as a strength, not a limitation: our Acetobacterium woodii CDMO programs are designed around the feedstocks the organism actually handles well—especially H₂/CO₂, formate, and methanol—so development begins with biological truth instead of marketing fiction.
The host is also increasingly engineerable. Recent work has expanded the genetic toolbox for A. woodii, including electrotransformation methods, endogenous CRISPR-based engineering, and metabolic rewiring studies that redirect electron flow or enable non-native product formation. That matters because it shifts the organism from a research curiosity to a programmable manufacturing chassis. Native acetate formation remains central, but the platform now credibly supports engineering toward products and intermediate chemistries beyond acetate alone.
Platform logic: acetate first, advanced products second
A serious Acetobacterium woodii CDMO service does not begin by pretending every desired molecule should be produced directly at commercial titer in a first-generation strain. The most robust industrial logic is usually acetate-first platform design: use A. woodii to convert carbon-efficient C1 feedstocks into acetate, formate, or other accessible intermediates with high selectivity, then decide whether the business case supports direct product extension, co-culture, or a staged downstream conversion process. Reviews of acetogenic gas fermentation and integration schemes continue to reinforce the value of this architecture.
That does not mean the platform is limited to acetate. Published engineering work in A. woodii has already demonstrated non-native or extended product routes including lactate, butyrate, and isopropanol, and has shown that electron-flow engineering can materially shift metabolic outcomes. Those results matter not merely as isolated achievements, but as proof that the host can be developed into a broader product platform when the program is built with proper redox, energy, and pathway logic.
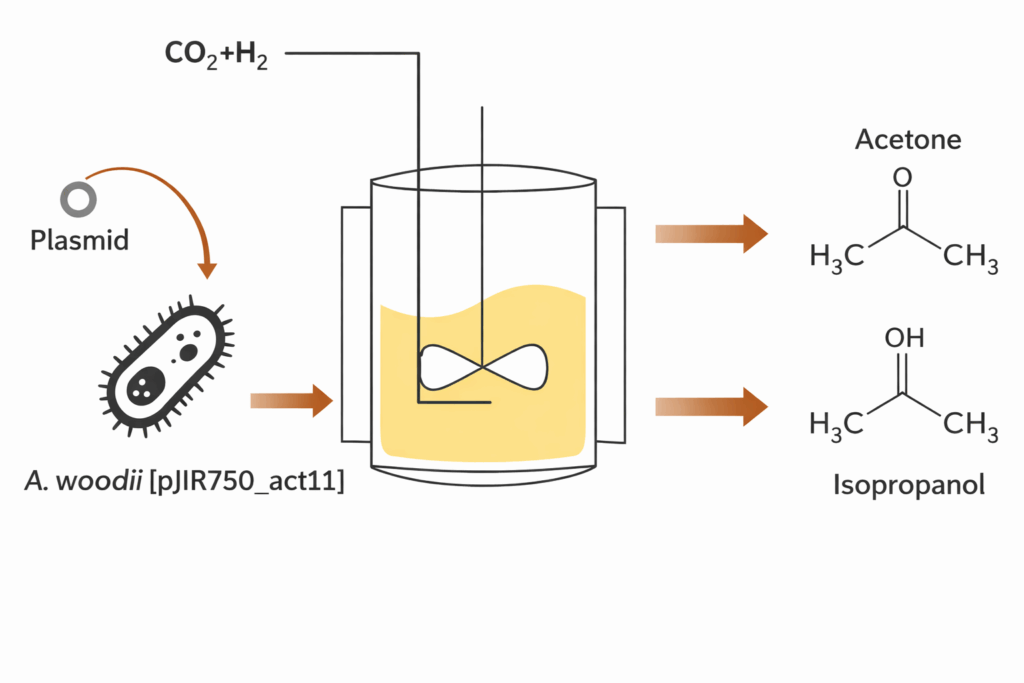
At Elise Biopharma, this becomes a structured development strategy. We map the sponsor’s QTPP to the host’s real biochemical operating envelope. If the target is bulk acetate, we optimize for gas uptake, sodium-balanced energetics, carbon efficiency, and downstream concentration. If the target is a non-native product, we define the redox burden, ATP implications, by-product profile, and process economics before scale-up begins. If the highest-value outcome is a staged process, we design Acetobacterium woodii contract manufacturing as the front-end carbon-fixation engine within a larger production system.
Strain engineering and cell bank strategy
Elise Biopharma’s Acetobacterium woodii CDMO services include strain qualification, bank creation, genotype confirmation, passage stability, and engineered-strain development under strict anaerobic handling. For native production programs, we begin with strain identity lock, purity, growth-characterization, sodium dependence confirmation, substrate utilization mapping, and baseline acetate productivity under defined gas and liquid feed conditions. For engineered programs, we layer in plasmid or chromosomal design, edit verification, phenotype confirmation, and stability profiling across serial passages.
This is where a great deal of hidden risk gets removed. In Acetobacterium woodii contract manufacturing, strain success is inseparable from operability. A line that performs in serum bottles but drifts under controlled gas feed, loses activity after passaging, or collapses when sodium balance shifts is not a production strain. Our onboarding workflow is built to prevent those failures from reaching pilot scale.
We characterize substrate response, sodium requirements, product spectrum, and growth under the exact control conditions that will matter later in steel or single-use anaerobic systems.
Upstream Process Development — Anaerobic Control as First Principle
Upstream work for Acetobacterium woodii starts with a non-negotiable constraint: oxygen must be completely excluded. This is not a tuning parameter—it is the foundation of the process. Every stage, from media preparation through inoculum expansion to final harvest, is designed to operate under strictly reduced conditions.
That means more than just inert gas blanketing. Media is conditioned to the correct redox potential, transfer paths are closed and pressure-balanced, and sampling systems are engineered to avoid even trace exposure. Small oxygen leaks or brief air contact events do not simply reduce yield—they can shift metabolism, damage enzyme systems, and create instability that only shows up later in the run.
Alongside oxygen control, redox stability and ionic balance are treated as primary variables. Shifts in electron availability, contaminant carryover, or ion composition can quietly move the system away from its optimal operating state. These effects are often subtle at first, but they compound quickly in acetogenic systems. The process is therefore built to maintain internal consistency, not just external control.
Gas and Feed Strategy — Matching Biology, Not Forcing It
Gas-fed fermentation introduces an additional layer of complexity. Performance depends on how well gas delivery aligns with metabolic demand, not on how much gas is pushed through the reactor.
Hydrogen uptake, carbon dioxide availability, and residence time are balanced against mixing and mass transfer so that the organism receives what it can actually process. Oversupplying gas does not increase productivity—it often leads to inefficiencies, stripping effects, or unstable conditions. The system works best when supply and consumption remain in equilibrium.
For liquid-fed approaches such as methanol or formate, the focus shifts to controlled input rather than maximum throughput. Feed rates are set to avoid toxicity, pH disruption, or redox overload. In more complex strategies—such as mixed feeds or staged transitions—carbon routing and electron distribution are mapped explicitly, ensuring the organism remains within a stable metabolic regime as conditions change.
Sodium Bioenergetics — Built Into the Process
A defining feature of A. woodii is its reliance on a sodium-ion gradient for energy conservation. This has direct consequences for process design.
Sodium concentration, ionic strength, and buffering systems all influence ATP generation and overall metabolic activity. If these parameters drift, performance follows. As a result, media composition is not treated generically—ion balance is maintained deliberately across development, scale-up, and production.
Rather than adjusting conditions reactively, the process is structured so that the organism’s energy system remains stable from the start. This creates a more predictable relationship between growth, substrate use, and product formation.
Downstream Processing and Product Recovery
Downstream handling in Acetobacterium woodii systems is less about purification in the traditional sense and more about managing carbon streams. The primary outputs are small molecules—most often acetate—rather than complex proteins.
This changes both the objective and the strategy.
Initial steps focus on stabilising the broth and removing biomass under conditions that preserve product integrity. From there, the path depends on the intended use of the product. In some cases, the goal is bulk recovery of organic acids using membrane systems, evaporation, or extraction techniques. In others, the broth is used directly as an intermediate for further chemical or biological conversion.
When processes are engineered toward alternative products—such as reduced compounds or short-chain molecules—recovery becomes more dynamic.
Volatility, solubility, and toxicity all influence how separation is handled. Techniques like gas stripping, solvent extraction, or adsorption may be introduced during fermentation itself to maintain stable production conditions. In these systems, recovery is not a final step—it becomes part of the process loop.
Chemistry Matters Early
One of the less obvious factors in successful recovery is upstream chemistry. Components introduced during fermentation—trace metals, buffering systems, reductants—carry through into downstream operations.
Carbonate buffering affects acid equilibria and determines how easily acetate can be recovered. Sulfur-containing compounds can interfere with catalytic upgrading. Even minor components can create separation challenges if they are not considered early. For this reason, medium design and downstream strategy are developed together, not in sequence.
When It Works
A well-aligned process behaves as a continuous system rather than a series of steps. Carbon flow, redox balance, and ionic conditions are matched to recovery methods so that the fermentation broth is already close to its final usable form.
When this alignment is achieved, separation becomes efficient, yields remain stable, and scale-up is predictable. When it is not, downstream becomes corrective—more complex, more expensive, and less reliable.
The difference is simple:
whether the process produces a mess to fix, or a stream ready to use.
Analytics, Quality, and Release Discipline
Analytical frameworks in Acetobacterium woodii programs are built to interrogate carbon flow, electron balance, and metabolic efficiency, not just endpoint concentration. The organism’s metabolism is tightly coupled to redox gradients and ion-driven energetics, which means that conventional titer-based metrics alone cannot explain process performance. A complete analytical picture must capture how substrates are partitioned, how electrons are distributed, and how energy conservation translates into product formation.
Core measurements typically include substrate uptake rates for hydrogen, carbon dioxide, methanol, or formate; product distribution across acetate and secondary metabolites; and intermediate pools such as acetyl-CoA-derived species. Beyond that, redox indicators—including NADH/NAD⁺ ratios, reduced ferredoxin levels, and hydrogen partial pressure—provide insight into pathway bottlenecks and metabolic saturation. These variables are especially important when pushing the system toward non-native products, where electron demand often exceeds native capacity.
Ion balance is another defining layer. Because A. woodii relies on sodium gradients for ATP synthesis, intracellular and extracellular sodium concentrations directly influence growth and productivity. Analytical programs therefore incorporate ion chromatography or equivalent methods to track sodium, potassium, and buffering species over time. Deviations in ion balance often precede visible drops in productivity, making them early indicators of process instability.
For engineered strains, genetic integrity and functional expression must be confirmed across scale. This includes sequence verification, stability of introduced pathways, and consistency of phenotype under production conditions. In acetogenic systems, phenotype drift may manifest not as loss of growth, but as subtle shifts in product spectrum or carbon efficiency—changes that only become visible through detailed metabolite profiling.
Gas-phase analytics are equally critical. Continuous monitoring of hydrogen uptake, carbon dioxide evolution, and off-gas composition provides a real-time window into metabolic activity. When integrated with liquid-phase data, these measurements allow full carbon and electron balances to be constructed, ensuring that process performance is understood mechanistically rather than inferred.
Release strategies are built around these same principles. Instead of relying solely on final product concentration, release criteria can include conversion efficiency, selectivity ratios, impurity thresholds, and stability under defined storage or transfer conditions. For processes feeding into downstream upgrading steps, consistency of intermediate composition is often more important than absolute purity.
The result is a data architecture that describes not just what the process produces, but how it behaves. That distinction is what allows acetogenic manufacturing to scale reliably—because variability can be traced to specific metabolic or physicochemical causes, rather than treated as unexplained noise.
Applications of Acetobacterium woodii CDMO Services
Acetobacterium woodii functions as a carbon-routing system—taking simple C1 inputs and turning them into structured, usable intermediates.
Core strengths:
- Converts H₂ + CO₂ → acetate with high carbon efficiency
- Handles methanol and formate as liquid feed options
- Produces clean intermediates for further upgrading
Primary applications:
• Acetate platform production
- Direct, efficient conversion via Wood–Ljungdahl pathway
- Stable supply of acetyl units
- Ideal starting point for downstream chemistry or biology
• Hybrid / multi-stage systems
- Acts as a front-end carbon converter
- Feeds second organisms or catalytic steps
- Enables chain elongation, alcohols, polymers, specialty molecules
• C1 feedstock utilisation
- Works with both gas and liquid carbon sources
- Flexible depending on cost, logistics, and infrastructure
- Simplifies integration into existing systems
• Engineered metabolite production
- Pathways can be redirected beyond acetate
- Supports formation of reduced compounds and short-chain products
- Requires tight control of electron flow and energy balance
• Carbon capture utilisation (CCU)
- Converts captured CO₂ into stable intermediates
- Links hydrogen-based energy systems to chemical production
Acetobacterium woodii Services — Top 30 FAQ
1. What is Acetobacterium woodii used for in manufacturing?
It is used to convert simple carbon sources like CO₂, hydrogen, methanol, and formate into acetate and other intermediates that can be further upgraded into fuels, chemicals, or materials.
2. Why use Acetobacterium woodii instead of other microbes?
It offers very efficient carbon utilization via the Wood–Ljungdahl pathway and can run on low-cost C1 feedstocks that many organisms cannot use effectively.
3. What products can be made with this platform?
Primarily acetate, but also engineered outputs like alcohols, organic acids, and intermediates for downstream bioprocesses.
4. Can it run on CO₂ as a feedstock?
Yes. It can fix CO₂ using hydrogen as an energy source, making it useful for carbon capture and utilization strategies.
5. Does it require oxygen?
No. It is strictly anaerobic—oxygen must be completely excluded throughout the process.
6. What makes the metabolism unique?
It uses the Wood–Ljungdahl pathway and a sodium-ion gradient for energy instead of the more common proton-based systems.
7. Is this platform scalable?
Yes, when gas transfer, mixing, and anaerobic control are properly engineered from early development.
8. What are the biggest technical challenges?
Maintaining strict anaerobic conditions, managing gas–liquid mass transfer, and controlling redox balance.
9. Can it use liquid feedstocks instead of gases?
Yes. Methanol and formate are commonly used alternatives that simplify handling compared to gas fermentation.
10. Is genetic engineering possible in A. woodii?
Yes. Modern tools allow pathway modification and redirection of carbon flux toward non-native products.
11. What industries benefit most from this platform?
Carbon capture, sustainable chemicals, biofuels, green manufacturing, and advanced materials.
12. How does it compare to yeast or E. coli systems?
It is slower-growing but far more efficient at converting simple carbon inputs into usable intermediates.
13. What is typically the first product made?
Acetate is the default output and often serves as a building block for further processing.
14. Can it be used in multi-step production systems?
Yes. It is commonly used as the first stage in processes that feed a second organism or catalytic step.
15. What kind of reactors are used?
Specialized anaerobic bioreactors with controlled gas input, mixing, and oxygen exclusion systems.
16. How is product recovery handled?
Depending on the product, methods include filtration, extraction, membrane separation, or direct integration into downstream processes.
17. What analytics are important for this system?
Carbon balance, gas consumption, metabolite profiling, redox state, and ion balance are all critical.
18. How stable are production strains?
With proper design and handling, strains can remain stable across multiple generations and scale transitions.
19. Is this technology ready for commercial use?
Yes. With proper process design, it can move from development to pilot and production scale reliably.
20. What makes Elise Biopharma strong in this area?
Deep focus on anaerobic systems, precise control of gas fermentation, and the ability to translate complex acetogenic biology into scalable manufacturing.
21. Why is this platform difficult for most CDMOs to execute well?
Because it’s not just fermentation—it’s anaerobic electrochemistry in a reactor. Gas transfer, redox balance, sodium gradients, and metabolic flux are tightly coupled. If one drifts, everything drifts. Elise treats these as one system, not separate problems.
22. How does Elise handle strict anaerobic requirements at scale?
Anaerobic control is built into every layer—media prep, transfer lines, reactors, sampling, and downstream handling. Oxygen isn’t “managed,” it’s designed out of the system entirely, including trace ingress points that typically get ignored.
23. What makes Elise’s gas fermentation approach different?
Gas delivery is engineered around biological demand, not equipment limits. Hydrogen uptake, CO₂ availability, and residence time are matched to metabolic rates so the organism never becomes substrate-limited or redox-stressed.
24. How does Elise manage redox balance in acetogenic systems?
Instead of reacting to redox problems after they appear, processes are designed around electron flow from the start. That includes feedstock ratios, hydrogen partial pressure, and pathway burden—so the system stays balanced under production conditions.
25. How are sodium-dependent energetics handled?
Sodium gradients are treated as a core process variable, not a media detail. Ion balance, buffer systems, and transport energetics are controlled to keep ATP generation stable throughout fermentation.
26. How does Elise approach scale-up differently for this organism?
Scale-up is based on mass transfer and metabolic equivalence, not just volume. Gas–liquid transfer rates, mixing profiles, and redox conditions are matched so the biology behaves the same at every scale.
27. What kind of niche feedstocks can be supported?
Beyond H₂/CO₂, processes can be designed around methanol, formate, hybrid feeds, or staged carbon inputs. The platform adapts to what’s most practical economically and logistically.
28. How does Elise handle non-native product engineering?
Pathways are introduced with full consideration of electron demand, ATP cost, and metabolic load. If the biology cannot support the target efficiently, the process is redesigned before scale-up—not after failure.
29. Can Elise support hybrid or multi-organism systems?
Yes. The platform is often used as a front-end carbon fixation step, feeding acetate or intermediates into a second organism or catalytic process. These interfaces are designed intentionally, not retrofitted.
30. How does Elise prevent common failure modes in acetogen projects?
By addressing them early:
- Gas limitation → solved through transfer design
- Redox imbalance → solved through feed strategy
- Product inhibition → solved through ISPR or staging
- Scale mismatch → solved through controlled scale-down models
Want to learn about Lactococcus? Click here: Lactococcus lactis CDMO Services
Conclusion
Acetobacterium woodii is not a plug-and-play organism—and that’s the point. It’s a specialist. When the process respects how it actually works—strict anaerobic conditions, sodium-driven energetics, and careful carbon flow—it becomes a surprisingly reliable way to turn simple inputs into useful outputs.
Think of it less like a traditional fermentation host and more like a carbon conversion engine. Feed it the right substrates, keep the environment stable, and it will consistently route carbon where you want it to go.
For teams working on CO₂ utilization, C1 chemistry, or multi-step production systems, it offers a practical and scientifically grounded way to move from concept to something that actually scales.
Email our team at info@elisebiopharma.com
