Built for Carbohydrate Precision, Immune Depth, and Product-Level Control
People often talk about glycoconjugates and glycoconjugate vaccine development as if they’re straightforward. They’re not, really.
The idea sounds simple enough—take a polysaccharide, link it to a carrier, and you get a better immune response. But once you actually start working with them, it becomes clear you’re not just linking two components. You’re shaping something that reflects every earlier decision, whether you intended that or not.
That’s usually where the tone of a programme starts to show.
At Elise Biopharma, we tend to work more deliberately from the beginning. The carbohydrate isn’t something we “fix later” if it causes trouble. The carrier isn’t chosen just because it’s familiar. And the chemistry isn’t there simply to make the linkage happen—it has to make sense for what the product needs to become.
Because once you’ve made the conjugate, it doesn’t hide much.
Why Glycoconjugate Vaccine Development Matters
If the structure is uneven, you see it. If the distribution is broader than it should be, it shows up in the data. If something wasn’t thought through early on, it tends to come back later—usually when timelines are tighter and options are fewer.
That’s really why this kind of work matters. It asks for a level of consistency that you can’t patch in at the end. The carbohydrate, the carrier, the chemistry—they all have to agree early, or the product never quite settles.

So we try to keep things aligned from the start. Not rigidly, just carefully enough that the work holds together as it moves forward.
When it works, the material feels more settled. It purifies without a fight. The analytics tell a clearer story. Scale-up doesn’t introduce surprises you then have to explain away.
That’s generally what we’re aiming for. Not something overdesigned. Just something that makes sense—and keeps making sense as the programme moves on.
What Glycoconjugate Vaccine Development Really Involves
A generic summary of glycoconjugate vaccine development might say: obtain the polysaccharide, activate it, conjugate it to a carrier protein, purify it, formulate it, and move into development. That is a nice sequence. It is not enough.
Real glycoconjugate vaccine development involves:
- carbohydrate source and quality strategy
- control of chain length and structural preservation
- O-acetylation or other structurally meaningful carbohydrate features
- activation chemistry
- carrier protein choice
- conjugation conditions and substitution control
- removal of free saccharide and unconjugated carrier
- analytical definition of the final conjugate population
- formulation and adjuvant compatibility
- scale-up behaviour and comparability planning
Every one of these can become a success point or a failure point.
At Elise Biopharma, glycoconjugate vaccine development begins by treating the polysaccharide as more than a raw antigenic input. We consider its length distribution, stability, modification state, and suitability for the intended chemistry. We treat the carrier as more than a plug-in helper, because its properties affect coupling, product heterogeneity, analytical behaviour, and long-term formulation logic. We treat the conjugation route as the place where the product becomes itself, which means the chemistry must be tuned to create a useful and reproducible result, not merely “some conjugate.”
This matters because glycoconjugate products often become difficult not because one single catastrophic mistake was made, but because the programme tolerated small ambiguities at several stages that later multiplied into a less manageable final product. That sort of drift is exactly what stronger glycoconjugate vaccine development work is designed to prevent.
One of the reasons we enjoy this area so much is that it rewards precision without becoming sterile. There is genuine scientific character to conjugate development. It is exacting, yes, but also rich. A good conjugate programme feels crafted in a way that many simpler products do not. And a great CDMO knows how to protect that craft while still building something scalable.
Carbohydrate Control Is Where the Product Story Begins
In glycoconjugate vaccines, the carbohydrate is not merely the antigenic input. It is an early determinant of the finished product. Chain length distribution, substitution pattern, structural integrity, and process sensitivity all influence what kind of conjugate can ultimately be made, how reproducibly it can be manufactured, and how clearly it can be characterized. That is why strong programmes do not treat the saccharide as upstream feedstock. They treat it as a core product variable from the first development decision onward.
At Elise, we approach the carbohydrate with that full product mindset. We evaluate source and process history, molecular size profile, activation susceptibility, lot-to-lot consistency, and structural features such as O-acetylation where biologically relevant. We also look closely at how those properties will behave under downstream processing stress, because a saccharide that appears acceptable in isolation can become problematic once activation, conjugation, purification, and formulation begin to apply real chemical pressure.
This matters because polysaccharide quality is not an abstract technical issue. It affects glycan epitope presentation, conjugation efficiency, free saccharide burden, and the long-term comparability story that regulators will eventually examine. A poorly controlled carbohydrate population can create broad heterogeneity in the final material, obscure analytical interpretation, and complicate process scale-up. A better controlled one gives the programme a clearer path to robust critical quality attributes, or CQAs, from early development through technology transfer.
That is one reason Elise builds from the assumption that saccharide properties continue to matter at every stage of development. They influence not just reaction performance, but purification strategy, analytical legibility, and the immunologic coherence of the final vaccine. In practical terms, this means controlling what many programmes leave vague until later: size distribution, chemical modification state, depolymerization risk, and compatibility with the intended conjugation route.
Weak programmes often underestimate the carbohydrate early and pay for that optimism later with unstable processes, unclear analytics, and difficult comparability exercises. Stronger programmes recognize that the saccharide is already describing the product before conjugation even begins.
Carrier Selection Is an Immunological and Chemical Design Decision
A conjugate vaccine is not a polysaccharide with a protein attached as an afterthought. It is a purpose-built molecular construct in which the carrier plays an active role in both chemistry and biology. Carrier choice influences T-cell help, substitution profile, steric accessibility, manufacturability, and the interpretability of the finished material. It also affects multivalent strategy, formulation behavior, and how prior carrier exposure may shape immune response.
That is why glycoconjugate vaccine development becomes much more sophisticated than simply choosing a familiar carrier and proceeding by convention. Familiarity can be useful, but it is not a sufficient design principle. The real question is whether a given carrier supports the product logic of the target vaccine: its intended immunological mechanism, its conjugation chemistry, its substitution tolerance, and its downstream analytical profile.
At Elise Biopharma, carrier selection is treated as a strategic design variable rather than a ceremonial choice. In some programmes, a classical carrier remains the best option because it supports a strong balance of precedent, scalability, and immune performance. In others, a recombinant carrier, alternative architecture, or more specialized design may create a cleaner process and a more controllable final material. The value lies not in choosing what sounds established, but in choosing what best supports product identity.
This distinction matters because carrier behavior can change the entire development pathway. Different proteins present different reactive handles, folding behaviors, and substitution tolerances. They may respond differently to activation conditions, generate different heterogeneity patterns, and require different purification logic after conjugation. In multivalent constructs, these effects become even more important because one carrier decision can ripple across several saccharide components and magnify complexity.
A disciplined programme therefore treats the carrier not as a passive scaffold, but as a central part of the vaccine’s chemical and immunological architecture.
Conjugation Chemistry Is the Point Where the Product Becomes Real
Much of the true difficulty in this field sits inside the chemistry. Conjugation is not just a linking step. It is the product-forming event. It defines how the saccharide is displayed, how much structural integrity is retained, how much heterogeneity is introduced, and whether the final construct will remain interpretable across analytics, stability studies, and scale-up.
At Elise, glycoconjugate vaccine development is built around respect for that reality. We support classical activation and linking strategies where they are scientifically appropriate, but we also recognize the value of more controlled approaches when the molecule demands it. The goal is not fashionable chemistry.
The goal is a chemically coherent, defensible, and scalable product.
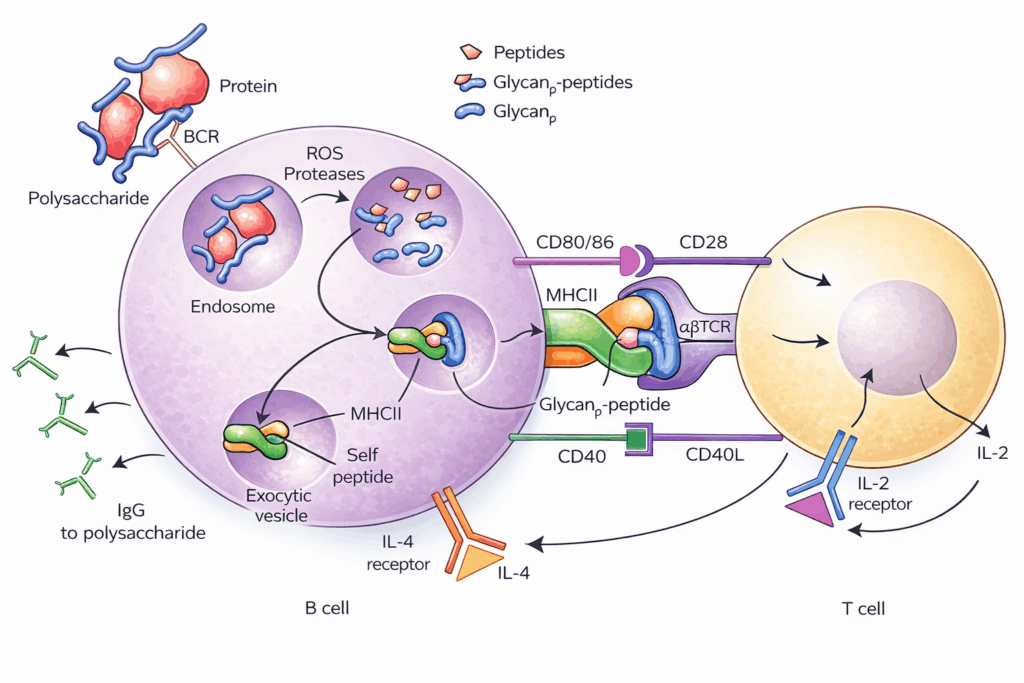
This is where concepts such as site-selective conjugation and controlled degree of substitution become important. A reaction that produces high apparent loading but poor structural coherence is not necessarily a success. If it broadens the product distribution, masks relevant epitopes, damages carrier identity, or creates an unstable purification problem, it may weaken the programme despite looking productive on paper. By contrast, a more disciplined reaction that yields a narrower and more reproducible conjugate population often creates much greater value over the life of the asset.
The chemistry must therefore be judged by more than first-pass coupling efficiency. It must be evaluated by what it does to product heterogeneity, free saccharide control, structural retention, lot consistency, and downstream analytical interpretability. A useful conjugation process is one that can be scaled without becoming mysterious.
Purification and Analytics Decide Whether the Molecule Can Be Defended
A conjugate that cannot be purified cleanly or characterized clearly is not a mature product. It is a difficult intermediate. This is why purification and analytics cannot be treated as downstream cleanup functions. They are part of the product design logic from the start.
Purification strategy must account for unreacted carrier, residual activated species, free saccharide, and broad molecular distributions that may emerge during conjugation. Analytical strategy must then be able to distinguish signal from noise with enough precision to support development, release, comparability, and eventual regulatory review. In practice, that can involve orthogonal methods such as SEC-MALS for size and distribution, HPAEC-PAD for saccharide characterization, and NMR or related structural tools where substitution state and integrity must be better understood.
The point is not to assemble the longest analytical menu. It is to build a characterization package that answers the right product questions. What is the conjugate population? How broad is it? How much free saccharide remains? Has an important structural feature been lost? Is the carrier still behaving as expected? Can the same material profile be reproduced reliably across lots?
At Elise Biopharma, we treat analytical legibility as a sign of product quality, not just documentation quality. A better product is one that can be read clearly by the methods that matter.
The Serious Standard in Conjugate Vaccine Design
The most credible conjugate programmes are built on agreement between carbohydrate, carrier, chemistry, purification, and product strategy. When those elements are aligned, the vaccine has a stronger chance of being chemically coherent, immunologically meaningful, operationally scalable, and regulatorily defensible. When they are misaligned, complexity accumulates quickly and usually reveals itself later, when correction is slower and more expensive.
That is the standard Elise Biopharma applies. We do not treat the conjugate as a byproduct of linking or as an output created by habit. We treat it as the product itself: a defined molecular system whose value depends on structural control, process logic, and analytical clarity from the beginning.
In this category, that is what serious work looks like.
Purification and Product Clarification in Glycoconjugate Vaccine Development
Once conjugation has occurred, the product still needs to become cleaner, clearer, and more physically coherent without losing what was just built. That is where purification enters as a major differentiator in glycoconjugate vaccine development.
Purification must often address:
- free saccharide
- unconjugated carrier protein
- reaction byproducts
- activated residual species
- broad product distributions
- aggregation or high-mass material
- formulation-ready buffer transitions
At Elise Biopharma, purification in glycoconjugate vaccine development is not a standard polishing step. It is one of the places where product identity is either preserved or blurred. A purification train that removes the right impurities but also destabilises the conjugate, selectively enriches the wrong fraction, or complicates the analytical story is not a strong purification train.
That is why we design purification around:
- what the final product must be
- which impurities truly matter
- how the conjugate population behaves physically
- what downstream analytics need to see
- what formulation and scale-up will later require
This is another place where strong glycoconjugate work earns its reputation. Conjugates rarely forgive crude downstream thinking. They reward processes that are selective, calm, and structurally informed. Elise Biopharma is built for that kind of work.
Analytics in Glycoconjugate Vaccine Development
There are very few product classes in vaccines where the analytical burden is as revealing as in glycoconjugate vaccine development. That is not a weakness. It is a sign that the product is sophisticated enough to require proper explanation.
At Elise Biopharma, analytics for glycoconjugate vaccine development may include:
- identity work on both carbohydrate and carrier
- free saccharide analysis
- degree of substitution analysis
- chain length distribution studies
- structural modification tracking such as O-acetylation where relevant
- conjugation efficiency assessments
- aggregation or size-distribution measurements
- potency or biologically relevant readouts
- stability-linked product drift monitoring
These methods matter because a conjugate is not fully understood through a single assay. A product can be “there” and still not be the right product. It can pass identity and still be too heterogeneous. It can look fine in release and still create comparability pain later. The analytical package in glycoconjugate vaccine development must therefore do more than verify existence. It must help define and protect product meaning.
This is one of the strongest areas for building authority online as well. A page like this communicates that Elise understands the burden of explanation that conjugates impose, and is comfortable meeting it.
That matters to technically literate buyers. It also happens to matter to regulators.
Scale-Up, Comparability, and the Long Life of a Conjugate Program
A conjugate product does not stop developing once the chemistry works. In many ways, it begins to reveal its real nature only when the process starts to scale, shift, or move.
Scale-up in glycoconjugate vaccine development can affect:
- carbohydrate processing consistency
- activation behaviour
- substitution patterns
- product distribution breadth
- impurity clearance
- formulation fit
- stability behaviour
- the interpretability of the analytical package
That is why Elise Biopharma builds glycoconjugate vaccine development with a strong view toward comparability and future process changes. A product that is exciting in early development but intolerant of normal manufacturing evolution is not as strong as it first appears.
The best conjugate programmes are the ones that can survive:
- scale increase
- process refinement
- site transfer
- raw material adjustments
- formulation optimisation
- multivalent integration
Without losing their identity or becoming analytically chaotic.
This is where Elise’s integrated model becomes especially powerful. We do not isolate development from later product life. We build the chemistry, purification, analytics, and formulation with the assumption that the programme will want to grow. A conjugate that cannot survive growth is not truly mature.
Examples of Where Glycoconjugate Vaccine Development Changes the Outcome
A few representative examples illustrate the value of true glycoconjugate vaccine development discipline.
Example 1: Strong Antigen, Weak Product Distribution
A sponsor begins with an attractive carbohydrate target and familiar carrier, but the resulting conjugate population is too broad and difficult to interpret. Better control of chain length and substitution logic narrows the product, improves analytical clarity, and makes the programme far more scalable.
Example 2: Familiar Carrier, Difficult Chemistry
A classical carrier is selected early for comfort and precedent. The chemistry works, but the product is noisier than expected and purification becomes burdensome. A more thoughtful carrier-and-chemistry pairing creates a cleaner final vaccine and a calmer downstream path.
Example 3: Scale Reveals the Real Product
An early conjugate programme looks fine until process scale-up subtly shifts carbohydrate behaviour and product distribution. Because the work was framed inside a real glycoconjugate vaccine development mindset, the structural source of the issue is recognised and addressed before it becomes a late-stage comparability crisis.
These are exactly the sorts of situations that separate a broad vaccine manufacturer from a true glycoconjugate specialist.
Why Elise is the best in Glycoconjugate Vaccine Development
Many companies can say they handle conjugates. Far fewer can support glycoconjugate vaccine development with true depth across carbohydrate handling, carrier logic, chemistry, purification, formulation, analytics, and process scale-up. That is why this page matters.
At Elise Biopharma, our strength lies in integration. We connect glycoconjugate vaccine development to:
- carbohydrate CDMO services
- carrier protein selection
- polysaccharide chain length analysis
- degree of substitution analysis
- vaccine potency and stability work
- formulation and adjuvant compatibility
- comparability and process development
That integrated framework matters because conjugates are one of the clearest examples of a product class that punishes fragmented thinking. A company that understands only the protein side will miss the carbohydrate. A company that understands only the carbohydrate side will miss the immunologic and manufacturing implications of the whole product. Elise is built to hold the full picture.

It also matters because glycoconjugate vaccine development remains one of the most credible categories in vaccinology. It combines technical richness, practical importance, and long-term market seriousness. A CDMO that can truly excel here signals something meaningful: not just capability, but discipline.
And discipline is what this product class respects most.
Conjugates Reward Precision
At Elise Biopharma, glycoconjugate vaccine development means designing vaccines as integrated products, not stitched-together parts. The carbohydrate, carrier, chemistry, purification, and product logic must work as one to create something clear, functional, and scalable. In this field, that is the standard that matters.
Contact our team at: info@elisebiopharma.com

