Adaptive CDMO Biologic Manufacturing: The Era Beyond Platforms
Biologic programs don’t all behave like monoclonal antibodies—and pretending they do costs time and money. This piece lays out a practical alternative: adaptive CDMO biologic manufacturing. In plain terms, it’s a way of working that matches the process to the molecule, not the other way around. It brings the same disciplined playbook to very different modalities—RNA CDMO, enzyme CDMO, fermentation CDMO, vaccine CDMO, exosomes CDMO, and more—so you can move from concept to regulated product with fewer resets and cleaner data.

Here’s the core idea. Platforms are useful scaffolds, but they’re rarely sufficient on their own. An adaptive operation starts from first principles (host, vector, kinetics, CQAs), instruments the work with robust analytics, and makes formulation decisions that reflect real logistics. That’s how you reduce CMC risk while accelerating milestones—whether you’re running IVT mRNA with LNPs, expressing enzymes in E. coli or Pichia, or standing up IVD-grade master mixes. The result is speed without theatre, dossiers that read cleanly, and scale-up that doesn’t unravel at tech transfer.
In the page that follows, we’ll map the approach across expression systems, analytics, and formulation; explain how it applies to RNA CDMO and adjacent programs; and show where it pays off commercially (time-to-milestone, cost of goods, and regulatory clarity). Then we’ll close with a straight answer: when an adaptive model is the right tool—and when it isn’t.
The end of the platform illusion
For a decade, we told ourselves a soothing story: if monoclonal platforms industrialised antibodies, surely everything else could be templated too. Reality—puckish as ever—declined the invitation. ADC CDMO programmes would not behave like naked IgGs; bispecific antibodies CDMO workflows wobbled under charge heterogeneity; scFv–Fc fusion proteins demanded inelegant buffer compromises that never appeared in the slideware; and plasmid development CDMO teams found themselves arm-wrestling with recalcitrant repeats, GC swamps, and stubborn topology. Meanwhile, exosomes CDMO cohorts discovered that isolation yields and EV identity metrics could pivot on minute changes in shear, membrane choice, or tangential-flow parameters—none of which respect “platform” dogma.
On the nucleic-acid front, RNA CDMO projects multiplied—and with them, lessons learned the hard way. IVT mRNA synthesis services CDMOs realised that dsRNA, capping ratios, and template impurities could sabotage potency long after a gel looked divine. Sponsors chasing sheer velocity discovered that LNP CDMO choices—ionisable lipid libraries, PEG-lipid ratios, and mixing regimes—were as consequential as the RNA itself.
Yes, strong CleanCap capability helps; yet a perfect cap atop mismatched UTRs or an ill-judged poly(A) tail still underperforms in vivo. This is precisely where adaptive CDMO biologic manufacturing earns its keep: integrating sequence design, IVT kinetics, and LNP physics into one coherent, testable hypothesis rather than a handful of disconnected workstreams.
In other words, molecules are individuals. Platform thinking remains useful scaffolding, but the building still needs an architect who understands the terrain.
The most interesting players today aren’t merely the biggest tanks or the keenest quotes; they’re the CDMOs fluent across modalities—able to pivot from E. coli CDMO services to Pichia CDMO secretion to cell-free protein synthesis CDMO for emerging therapeutic startups—without hammering square pegs into cylindrical chromatography columns. Consider a telling data point from a kindred outfit: a ~93% batch success rate achieved without engineering lots, credited to bespoke analytics and formulation.
The moral isn’t “platforms are dead”; it’s that platforms must be permeable, instrumented, and governed by adaptive CDMO biologic manufacturing principles. When the molecule misbehaves—as it often does—you need people who can read its tells, design the right experiments, and move decisively. That’s the new competitive axis, and adaptive CDMO biologic manufacturing is how you stay on it.
Biology rediscovers itself: the hemifusome as parable
Cell biology, bless it, just revealed a new organelle—the hemifusome—spotted with cryo-electron tomography, likely involved in protein sorting and recycling. It’s as if nature is reminding us: there are still rooms in the cell we haven’t walked through. What a deliciously humbling correction for manufacturing dogma.
Why does a nanoscale curiosity matter to a CDMO? Because the hemifusome embodies the logic we now need in factories: selective fusion (merge what must be merged), compartmentalisation (keep what must be kept apart), and efficient reuse (salvage value from every stream).
Modern microbial development and manufacturing services now look less like blunt production lines and more like living logistics—dynamic, instrumented, reconfigurable. The mindset that discovered a 100-nm structure with the right tool—and then asked better questions—mirrors the mindset that characterises the best RNA CDMO and oncolytic virus CDMO shops: curious, measurement-obsessed, sceptical of comforting assumptions.
The Rise of Adaptive Manufacturing — Integration Across Expression, Analytics, and Formulation
The shift toward adaptive biomanufacturing marks a fundamental rethinking of how modern organizations approach complexity in life sciences. Instead of dividing microbial, enzymatic, and nucleic-acid programs into rigid silos, adaptive operations bring these domains into a single, continuous system—linking expression, analytics, and formulation under one data framework. The result is a manufacturing model that replaces static, one-size-fits-all platforms with flexible, evidence-driven methods built on reproducibility and scientific rigor.
Microbial Systems: The Industrial Foundation
Microbial expression remains the backbone of modern biomanufacturing. E. coli leads where speed, cost, and yield matter most—especially for enzymes and research proteins. Codon optimization, signal peptide tuning, and periplasmic targeting are now essential for proper folding and function. What separates scalable operations from lab-scale mimicry is discipline in downstream control: endotoxin removal, host-protein clearance, and ion-exchange chromatography run with precision, not guesswork.
When secretion or glycosylation is critical, Pichia pastoris bridges bacterial efficiency with eukaryotic sophistication, producing secreted titers that simplify purification. For probiotics, food-grade enzymes, and live biotherapeutics, Bacillus remains the trusted GRAS/QPS workhorse. Adaptive manufacturers fluent in all three systems choose the host based on biochemical rationale and commercial logic—not convenience.

Function Before Appearance
True enzyme production is judged by function, not looks. Absorbance ratios and perfect gels are tools, not trophies. What matters is catalytic performance in real matrices, solvent tolerance under conjugation conditions, and mechanical stability through shipping and use. Experienced developers test immobilization chemistries—epoxy, aldehyde, ionic—and track reusability under process stress. Every step ties back to measurable activity and lifecycle cost. In adaptive operations, form follows function, and every molecule must earn its beauty through performance.
Nucleic Acids: The Discipline of Precision
Precision begins long before the reactor runs. In RNA and DNA manufacturing, design defines destiny. Template structure, promoter strength, and magnesium balance determine transcription yield more than any downstream correction. The smallest sequence decision echoes through every stage of production.
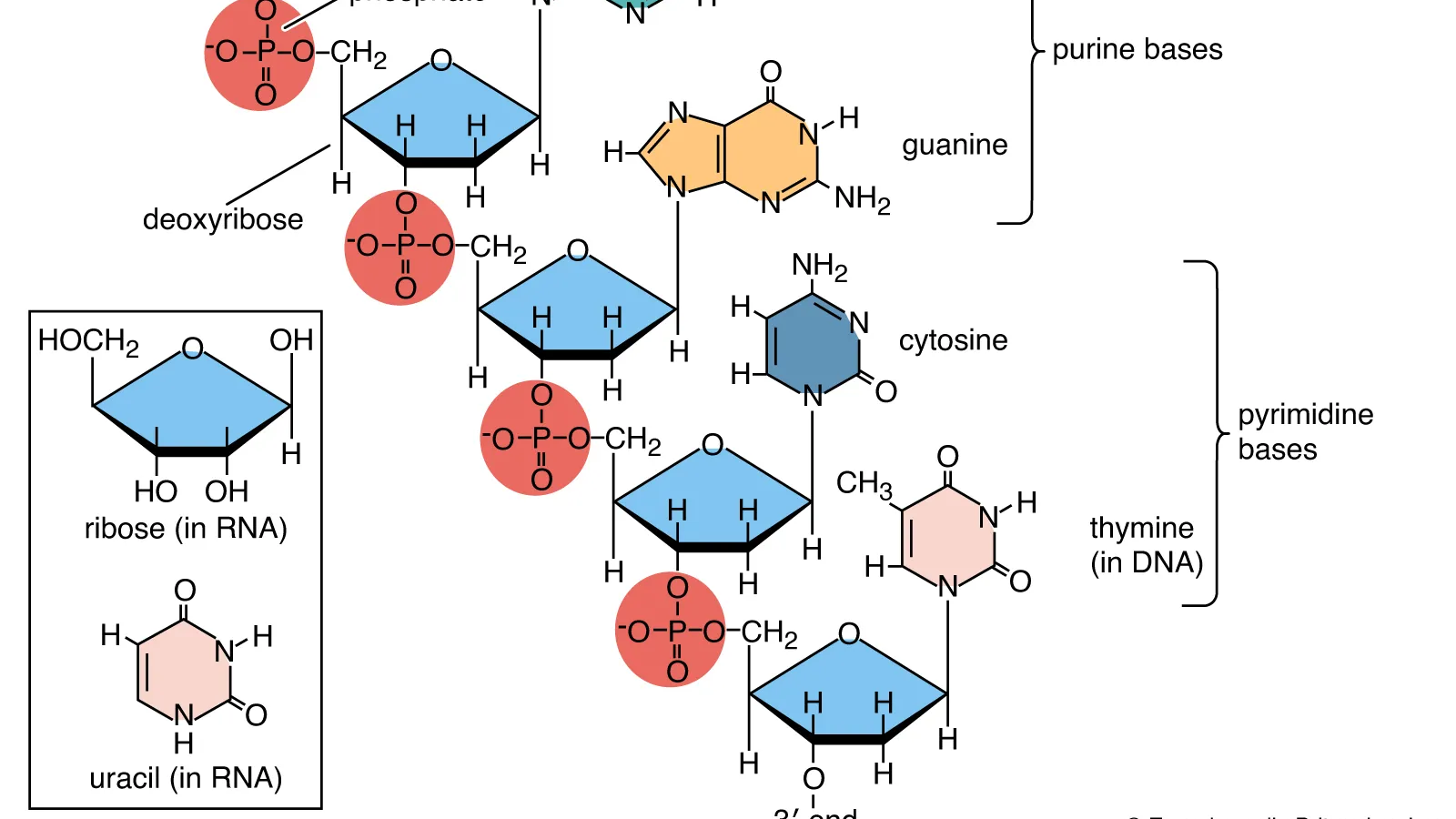
In adaptive systems, poly(A) tailing is an act of fine control—encoded or enzymatic, timed to balance translation efficiency and stability. Capping choices follow evidence, not tradition: co-transcriptional for speed, enzymatic when fidelity matters most. Double-stranded RNA is addressed at its source, removed before it can compromise potency. Each adjustment is purposeful, each improvement cumulative—the quiet craft of precision turning fragile strands into viable therapeutics.
The Architecture of Formulation
When RNA meets its lipid carrier, chemistry becomes engineering. Formulators tune lipid ratios like conductors, adjusting particle size and PDI to maintain structure and stability. Oxidation, temperature, and motion are modeled, not hoped for. The result is predictable shelf life and consistent potency—a molecular architecture designed to endure. Each batch becomes proof of discipline: stable, uniform, and ready for the journey ahead.
Expanding Frontiers: Vesicles, Phages, and Probiotics
Adaptive manufacturing is defined by curiosity. Beyond proteins and nucleic acids lie exosomes, phages, and probiotics—each demanding its own form of precision.
Exosomes require immaculate upstream hygiene and dual purification—SEC with AEX—to ensure vesicle integrity and purity. Phage therapeutics balance chaos with control: host-DNA clearance, endotoxin removal, and potency assays that treat disorder as data.
Probiotics complete the circle—living systems monitored like instruments. Strain identity is confirmed by sequencing, viability mapped across storage conditions, and stability modeled under real-world pressure.
Together, these domains show how adaptive manufacturing transforms biology into an ecosystem of intention—where every cell, particle, and vesicle is handled with both technical clarity and quiet respect.
Learn more about our Phage CDMO partner Phage Biologics
Diagnostics, Veterinary, and the Convergence Ahead
Adaptive manufacturing stretches easily across boundaries. In diagnostics, reproducibility becomes the love language of reliability — every reagent designed to survive transport, storage, and contact with complex biological matrices. In veterinary biologics, the rhythm changes: cost efficiency meets microbial craftsmanship, building affordable vaccines, enzyme supplements, and microbial therapies that still meet rigorous safety and efficacy thresholds.
This convergence — of disciplines, domains, and demands — is no accident. It is the natural evolution of systems that think in loops rather than lines, treating biology not as a series of problems but as a dynamic conversation between design and response.
The Constant Principle
Across every modality—whether protein, nucleic acid, or live-cell platform—the same governing truth applies: sustainable success depends on combining flexibility with discipline. Adaptive biomanufacturing is not about perpetual improvisation; it is a structured framework for intelligent adjustment, where variability is anticipated, controlled, and—when advantageous—leveraged.
At its core, adaptive manufacturing converts experimentation into intelligence. Expression data inform analytical design; analytical results shape formulation; formulation feedback drives process refinement. Each loop strengthens reproducibility while revealing the true performance boundaries of a molecule.
In this context, adaptability is not the absence of standards but their strategic application. The objective is to maintain precision under changing biological, operational, and regulatory conditions—whether optimizing an RNA template, balancing enzyme kinetics, or stabilizing complex bioconjugates.
The most advanced organizations demonstrate several defining characteristics:
- Data-driven responsiveness: real-time process monitoring and statistical control that allow proactive correction rather than reactive troubleshooting.
- Integrated development: expression, purification, and formulation designed as a single continuum rather than discrete functions.
- Regulatory fluency: documentation and validation plans built to satisfy auditors without impeding innovation.
- Operational maturity: the ability to flex workflows for discovery programs while maintaining GMP-aligned rigor for late-stage products.
Adaptive manufacturing, when executed properly, reduces uncertainty without diluting scientific integrity. It allows sponsors to progress from design to dossier with fewer discontinuities, faster learning cycles, and clearer decision points. The result is not improvisation disguised as innovation, but discipline refined by feedback—a modern equilibrium between control and evolution that defines the future of biologics manufacturing.
The circular bioprocess: fermentation as ecology
A shift is under way—from “can we?” to “how shall we?” It shows up in conference corridors: fewer debates about possibility, more arguments about optimisation. In fermentation and downstream, that means process water reclamation, energy-aware CIP, smarter UF/DF staging, and computational mass-balance sanity checks. In microbial development and manufacturing services, adaptive fed-batch and perfusion are less about novelty and more about duty cycles and space-time yields. In ion exchange chromatography CDMO services, step-elution vs gradient trade-offs now factor resin cost curves and raw-material lead times, not merely purity.
Zoom out and a pattern emerges. The best programmes treat the facility as an organism—inputs, outputs, and homeostasis tracked and tuned. The circularity isn’t ideology; it’s economics.
The immunological frontier: iPSC and the new GMP
Cell therapy keeps teaching us that biology’s versatility can be industrialised—but only with unromantic discipline. iPSC-derived CAR-immune cells (T, NKT, macrophages) are demonstrating batch-to-batch uniformity that autologous systems can barely dream of. The manufacturing pipelines—xeno-free, feeder-free, tiered banking (Seed→MCB→WCB)—feel like 21st-century riffs on old vaccine logic, now enforced by sequencing, karyotyping, and multi-omic sanity checks.
For CDMOs, the implication is clear: potency assays must mirror mechanism (phagocytosis for CAR-macrophages; cytokine harmonics for immunomodulators), release panels must be brutal (sterility, mycoplasma, residuals), and analytics must be narratively coherent—i.e., defensible to regulators without baroque rationalisations. If that sounds like in vitro biology CDMO services, in vitro biology and screening CDMO, and CDMO partner for diagnostics fusing into one practice, that’s because they are.
Customization isn’t vanity—it’s risk control
Forcing every molecule through the same template is how timelines slip. The faster, safer path is to face a program’s quirks early—with targeted experiments, honest data, and quick iterations.
What to tailor:
- Assays & formulations. Tune tests to the biology and choose excipients that survive real logistics.
- Analytical depth. For hybrids (e.g., small-molecule conjugates), confirm solid forms and charge variants up front. For fusion proteins, map glycans before they surprise you at scale. For plasmids, design to avoid slippage and topology headaches. For phage tools, track host DNA and endotoxin with the same discipline you’d bring to viral vectors.
What to standardize:
- Documents & human factors. Keep SOPs, data models, training, and batch records boringly consistent. Minimize variation everywhere except where the molecule demands it.
That’s the balance: customize where it reduces risk, standardize where it reduces noise.
The next advantage: a “fractal” manufacturing model
The durable edge won’t be one shiny technology; it will be an operating shape—self-similar from bench to plant, adaptable at every handoff.
Core elements:
- Common spine, local nuance. Shared quality systems, data schemas, and training across sites; host- and region-specific playbooks where needed.
- Continuous sensing. Real-time PAT for upstream and downstream so drift is caught before it becomes deviation.
- Smarter experimentation. Design tools that propose the next perturbation based on live data, not last quarter’s averages.
- Tight joins. Clean handoffs between nucleic-acid production and delivery (e.g., IVT to LNP), and between analytics and formulation.
- Supply foresight. An early-warning layer for resins, lipids, enzymes, and single-use so schedules don’t hinge on luck.
Optional “boosters” fit naturally into this geometry—endotoxin-free expression, glycan mass-spec suites, synthetic DNA services—without breaking the system’s rhythm.
In short: build a network that behaves like biology itself—diverse enough to be resilient, rule-bound enough to be predictable.
What this means for sponsors (and how to choose partners intelligently)
You don’t need a cathedral; you need a campus. When surveying the field—be it for radioconjugates CDMO, exosomes CDMO, ADC CDMO, mRNA synthesis services CDMO, enzyme CDMO, or fermentation CDMO—benchmark partners on five very human questions:
- Will they measure what matters?
For RNA CDMO: integrity by capillary methods, cap structure by LC–MS, dsRNA orthogonally, potency in relevant cells, lipid degradants for LNPs, and stability in the actual shipping profile.
For vaccine CDMO and protein vaccines CDMO: antigen integrity, epitope preservation, adjuvant compatibility, and field-reconstitution kinetics.
For ADC CDMO: DAR distribution, charge variants, linker stability, and formulation stress. - Can they make and break platforms gracefully?
Reuse proven playbooks for speed; deviate without drama when the molecule insists. That includes swapping fermentation hosts (hello, Pichia CDMO), pivoting from batch to perfusion, or embracing cell-free protein synthesis CDMO when time-to-grams trumps everything. - Are they eloquent in downstream?
Not just “we have columns” but we model mass transfer, match membranes to aggregation behaviour, and choose gradients with resin economics and lead times in mind. Ion exchange chromatography CDMO services should sound like a craft, not a catalogue. - Do they treat logistics as part of the molecule?
Rapid turnaround lyophilization services are fabulous—if the lyophilisate survives humid customs warehouses. Good partners design for the real world, including “ambient” success when appropriate. - Do they write dossiers you’re proud to submit?
IND/CTA/IMPD packages that read like the science—clear, justified, reproducible—predict smooth reviews. The opposite foretells month-eating clarifications.
A Note on Elise Biopharma’s Place in This Landscape
Elise Biopharma sits squarely at the center of adaptive CDMO biologic manufacturing—a philosophy that treats manufacturing not as a checklist but as a living science. The company was built on a microbial-first foundation, matured through enzyme expertise, and evolved into an RNA-fluent, diagnostics-savvy, and formulation-realistic CDMO that thrives on precision and adaptability. Its culture—half Cambridge, Massachusetts rigor, half Montréal inventiveness—embodies what adaptive CDMO biologic manufacturing actually means in practice: curiosity paired with control, innovation anchored in compliance.
At Elise, designed experiments are muscle memory, not theatre. The team builds DOE matrices as naturally as others write SOPs. Quality is scaled to context—from RUO agility to ISO 13485 traceability and full GMP alignment—ensuring each molecule receives the level of scrutiny it deserves. The essence of adaptive CDMO biologic manufacturing here is simple: never overbuild process where science suffices, and never underpower science where regulation demands robustness.
Microbial and Enzyme Depth
Elise’s microbial division defines its roots in adaptive CDMO biologic manufacturing. The group leverages E. coli CDMO services for rapid expression campaigns, balancing high titers with refined refolding strategies and endotoxin-aware DSP. For secreted proteins, Pichia CDMO systems extend reach into glycoprotein territory—providing secretion, folding fidelity, and scale flexibility that bacterial systems cannot match.
Downstream, ion exchange chromatography CDMO services are treated as design spaces, not static recipes. Here, adaptive CDMO biologic manufacturing translates to data-driven tuning of resins, gradients, and buffer chemistry until purity, recovery, and economics align.
Downstream as a Design Space
In adaptive biomanufacturing, downstream processing is not a static sequence—it’s a design space governed by data. Ion-exchange chromatography, rather than a fixed recipe, becomes a field of fine adjustments: resin selection, gradient slope, and buffer composition tuned until purity, recovery, and economics reach equilibrium. Enzyme analytics follow the same discipline. Catalytic efficiency (kcat/Km), process-stress stability, and activity retention matter more than optical clarity or gel aesthetics. Precision is measured by reproducibility and functional truth, not appearances.
RNA Discipline
Adaptive manufacturing finds its purest test in RNA production. The philosophy integrates chemistry, biology, and logistics into a single feedback loop. It begins upstream—confirming template topology, promoter fidelity, and plasmid integrity before transcription even starts. Double-stranded RNA is minimized at its source, not scrubbed as an afterthought. Analytical confirmation follows: LC-MS for capping validation, poly(A) tail distribution profiling, and residual DNA quantification as routine checkpoints.
Formulation completes the cycle. Adaptive RNA programs treat solvent exchange, lipid degradant monitoring, and encapsulation efficiency as interdependent levers. LNP development links particle size, PDI, and potency to controlled process variables, ensuring reproducibility across scales. Storage format—whether cryogenic, refrigerated, or lyophilized—is chosen based on logistics and thermal data, not laboratory convenience. The discipline lies in treating formulation as engineering, not artistry.
Diagnostics and Veterinary Applications
Adaptive biomanufacturing extends beyond therapeutics. In diagnostics, the same operational rigor applies: reagents built to withstand matrix inhibition, master mixes qualified to ISO 13485, and documentation structured for regulatory transparency. Each assay behaves predictably under variable sample types and storage conditions because design begins with real-world constraints.
Veterinary biologics apply similar logic on different timelines. Programs prioritize rapid turnaround, rugged stability, and ethical cost efficiency. Adaptive teams integrate microbial expression, fermentation, and formulation strategies tuned for animal-health markets, where the economics and regulatory expectations diverge from human medicine. Cross-sector fluency enables data, processes, and standards to move fluidly between domains without dilution of quality.
Fermentation and Formulation Without Illusion
Adaptive thinking begins with physics. Fermentation scales by preserving oxygen transfer (kLa) and shear conditions that protect cellular health. PAT systems monitor metabolism in real time, ensuring consistent biomass and folding fidelity. The outcome is quiet reliability—processes that run without drama and products that behave as predicted.
Formulation follows the same pragmatic ethos. Freeze-drying is engineered around collapse temperature, glass transition, and reconstitution kinetics. Each variable—baffles, agitation, feed rates, drying steps—is a dial in an optimization loop, not a fixed parameter. The goal is repeatable performance under field conditions, not slide-deck promises.
Edge Cases Treated as Core Science
Adaptive manufacturers don’t avoid complexity—they systematize it. Phage programs, for instance, operate with validated bioinformatics tools tracking host-DNA carryover, potency, and endotoxin clearance at audit-ready precision. Exosome workflows embed vesicle biophysics from the start, balancing yield and RNA-cargo integrity with orthogonal analytics rather than nominal purity claims.
Even live-attenuated vaccine lyophilization follows this rigor: cycle development tied to product-temperature mapping, residual-moisture profiling, and cake morphology studies designed for global transport conditions. The defining trait is composure amid heterogeneity—treating every modality as a system to be understood, modeled, and improved, not a problem to be forced through a platform.
In the adaptive paradigm, biology is not tamed by templates but guided by understanding. The goal is control without rigidity—processes that evolve with molecules, markets, and time.
Top 10 FAQ: Adaptive CDMO Biologic Manufacturing
1. What exactly is an Adaptive CDMO?
An Adaptive CDMO (Contract Development and Manufacturing Organization) is a next-generation partner that tailors process design, analytics, and scale to each molecule’s biology instead of forcing it into rigid templates. It blends scientific flexibility with regulatory discipline—responding to real-time data, not outdated assumptions.
2. How does an Adaptive CDMO differ from a traditional CDMO?
Traditional CDMOs rely on predefined “platform” templates—great for monoclonal antibodies, poor for complex or novel modalities. Adaptive CDMOs, by contrast, use modular workflows, digital twins, and design-of-experiments (DoE) frameworks that evolve as data emerges. They prioritize outcome-based manufacturing rather than procedural uniformity.
3. Which molecule types benefit most from Adaptive CDMO biologic manufacturing?
Complex modalities—like bispecific antibodies, antibody-drug conjugates (ADCs), mRNA, enzymes, exosomes, and live biotherapeutics—gain the most. Each presents unpredictable kinetics or stability challenges that adaptive approaches can solve through customized fermentation, purification, and formulation strategies.
4. What technologies enable Adaptive CDMO biologic manufacturing?
Continuous monitoring (PAT), high-throughput analytics, AI-driven bioprocess modeling, and digital QMS systems make adaptive manufacturing possible. Tools such as LC-MS, capillary electrophoresis, and NGS are integrated into closed-loop control systems that learn and optimize every run.
5. Does Adaptive CDMO biologic manufacturing cost more?
Initially, adaptive CDMOs may seem pricier due to deeper development work. However, the approach reduces failure risk, batch rework, and regulatory remediation—cutting overall COGS and time-to-market. Smart design saves exponentially once the molecule reaches clinical scale.
6. How does regulatory compliance fit into an adaptive model?
Adaptive doesn’t mean experimental. Leading Adaptive CDMOs maintain phase-appropriate systems: RUO agility early, GMP-aligned controls later. Documentation, method validation, and comparability studies are built into the feedback cycle, ensuring every pivot is traceable and compliant.
7. What analytical strengths define an Adaptive CDMO?
Adaptive CDMOs treat analytics as the nervous system of the operation. They perform deep characterization—potency, stability, dsRNA levels, impurity mapping—and connect those results directly to process control. Release specifications reflect function, not vanity metrics.
8. Can Adaptive CDMOs handle multiple modalities under one roof?
Yes—and that’s their hallmark. A true Adaptive CDMO biologic manufacturing partner can pivot between E. coli microbial systems, Pichia secretory hosts, mRNA IVT suites, enzyme production, and lipid nanoparticle (LNP) encapsulation—all governed by unified data standards and cross-trained scientists.
9. How does adaptive philosophy influence formulation and fill-finish?
Adaptive CDMOs design formulations around logistics realities—cryogenic, refrigerated, or lyophilized—depending on regional distribution constraints. They use DoE to balance stability, potency, and reconstitution speed, ensuring the product survives the real world, not just the lab.
10. Why choose Elise Biopharma as your Adaptive CDMO partner?
Elise Biopharma represents adaptive manufacturing in its purest form: microbial-first roots, enzyme precision, RNA fluency, and diagnostic pragmatism—all unified under robust quality systems in Cambridge, MA and Montréal, Canada. It’s science with flexibility, compliance with creativity, and delivery without drama.
The Adaptive Conclusion
Adaptive biomanufacturing isn’t a brand posture; it’s a systems approach to reducing risk under biological uncertainty. The core idea is simple: link expression, analytics, and formulation in tight feedback loops, and let data—not templates—decide the next move. In practice, that means phase-appropriate controls, method validation that survives inspection, and supply chains modeled as part of the process rather than an afterthought.
A genuinely adaptive organization shows a few consistent traits:
- Permeable platforms: standardize data models, SOPs, and training; customize assays, unit operations, and formulations where the molecule demands it.
- Upstream-first quality: design DNA/RNA templates, hosts, and media to prevent defects (dsRNA, misfolding, glycan drift) rather than polishing them downstream.
- Continuous sensing: real-time PAT across upstream and downstream, with analytics treated as strategy—not paperwork.
- Transparent trade-offs: explicit choices on capping vs. co-transcriptional methods, poly(A) strategies, excipients, and cold chain, tied to target product profile and cost-of-goods.
Many companies—Elise Biopharma among them—offer concrete examples of this mindset in action, but the lesson is broader than any single provider. Adaptive operations scale from startup speed to BLA rigor because they make variation visible, controllable, and, when useful, intentional. If your program lives outside the monoclonal comfort zone (enzymes, RNA, exosomes, phage, probiotics, complex conjugates), an adaptive model turns complexity into a managed variable rather than a scheduling surprise.
In the end, “adaptive” is a commitment to first principles with modern instrumentation: design experiments that answer real risks, instrument the process so drift is caught early, and document decisions so regulators can follow the logic. Do that consistently, and molecules behave more like products—and launches look less like cliff dives and more like planned deliveries.
Contact our team directly at info@elisebiopharma.com
Learn more about –> Tangential Flow Filtration
Or learn about –> The Convergence of AI and Biopharma Data Analysis

